The Enzymes
Description
This section is from the book "Chemistry Of Chelation In Cancer", by Arthur Furst. Also available from amazon: Chemistry Of Chelation In Cancer.
The Enzymes
Functions of these trace elements and their relationships to enzymes are being extensively studied (92,268, 342, 560,612). More and more enzymes are being found to be metal-dependent or metal-containing (559, 604), and almost all models suggested to date involve the phenomenon of chelation of the metal by the enzyme. That only a small molecular portion of the enzyme protein is associated with the metal can be deduced from the knowledge of the structure of catalase. Only one atom of iron with an atomic weight 55.8 is bonded by one molecule of catalase having a molecular weight of 225,000. It must be recognized at the onset that chelate stability is not the only important consideration in metal enzyme complexes. Magnesium ion does not form stable complexes in the classical sense; yet this cation is a cofactor for more enzymes than any other element.
The zinc-containing enzymes may be quite important for growth of cancer cells, especially in the prostate; recently a new zinc requiring alkaline phosphatase was found in the ventral lobe of the rat prostate gland. Addition of EDTA inhibited the activity of this enzyme by 30-50%. Simultaneous addition of zinc and magnesium inhibited serum alkaline phosphatase activity, an effect more pronounced in cancer patients than in normal controls.
Non-essential trace metals may also influence enzymatic reactions. Thallium in the trivalent, not monovalent, state is an uncoupling agent, yet it did not depress oxygen uptake during the oxidation of glutamate by mitochondria; cadmium at the same concentration did inhibit oxygen uptake. Some elements, including arsenic, bismuth, iron, gold, silver and mercury, deposit in skin and displace deposited copper, releasing it to activate more tyrosinase. Non-essential metals can, therefore, act as enzyme stimulants and increase the kinetics by displacing the essential metals from tissues. In other cases the non-essential cation can possibly displace the needed metal from enzyme systems. This latter pathway has been suggested to account for the increase of cholesterol synthesis in the presence of chromic ion. Metal ion catalyzed reactions can be used as models for enzyme systems, especially for rate studies. Peptidases are enhanced by magnesium, manganese, zinc, cobalt and iron. The metal may act to bind the substrate to the enzyme (XC). The order of activity of metals in model experiments for enzyme activation may be the same as that previously given as the order of stability constants between ligands and cations. See page 12.

Chelating agents themselves may activate enzymes. Preparations of succinoxidase in an unfavorable environment like bicarbonate buffer (rather than phosphate buffer) are activated ten-fold by addition of either EDTA or 8-hydroxy quinoline; amino acids like lysine may also act as activators. Chelating agents as enzyme inhibitors are also known. Some metals increase toxicity of chelating agents if motility of spermatozoa is used as a test system.
Two types of metal-binding to enzymes can be recognized. The first requires a rather loose association between the metal and enzyme, the metal merely serving as an activator. In this case more than one metal may have similar activities; quite often magnesium-activated enzymes act just as well when manganese ion is added to the reaction medium. These elements have similar outer electron distributions. Either, as divalent cations, can activate any one of about thirteen enzyme systems, including deoxyribonuclease, phosphatases, arginase and cholesterol-synthesizing systems.
Metallo-enzymes, wherein a specific metal is attached to and isolated with a protein, constitute the second group. These enzymes differ in their activity after treatment with a chelating agent. These agents may remove the metals in some enzymes but not in others, or these agents may attach themselves to the metal without removing it.
Metals as part of enzymes can have a variety of functions. In addition to complexing with enzymes, essential metals may displace ineffective ions from active centers or can remove inhibitors. They may serve as the site for binding substrates and thus become themselves the active centers of the enzyme. The cation may first complex with the substrate and help establish the proper conformation for the eventual binding to the enzyme (XCI).
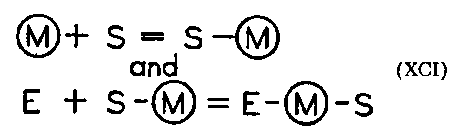
These metals may link the coenzymes to the enzymes (XCII), or may serve to keep two proteins together in a fixed geometric form, thus possibly helping to keep the correct coil.
A possible contribution to stereo specificity of enzymes can be made by the metal in that chelates exhibit optical and cis-trans isomerism. An example may be ferrous iron complex in protoporphyrin, called heme, which is found in peroxidase, catalase, cytochromes, myoglobin and hemoglobin. Four of the six bonds of iron are coordinated in a plane. The two additional positions may be coordinated perpendicularly (XCIII). These positions may be occupied by histidine which in turn attaches to a polypeptide; this results in catalase (XCIV).

The metal may act as a Lewis acid and reduce the electron density of the bond to be cleaved in the substrate. The cation, alternatively, may be the first point of contact, or be the means for the attraction of the hydroxyl ion which will attack that weakened bond.
Finally, the metal may serve an oxidation-reduction function, providing the metal can exist in more than one oxidation state under physiological conditions. The substrate may reduce, and an electron receptor may oxidize the metal, and thus a steady state may be achieved. Copper conceivably may function as a cuprous-cupric couple and be the electron carrier in Cytochrome A.

Electron paramagnetic resonance (EPR), or as some prefer, electron-spin resonance (ESR) is the newest tool for the investigations of changes in oxidation states of metals associated with enzymes. The activity of copper in enzyme systems can now be conveniently followed, for the paramagnetic cupric ion with one unpaired electron will give a responsive signal, whereas the dia-magnetic cuprous ion having only paired electrons will not. Differences have already been found in active and resting states of two copper-containing enzymes, fungal laccase and ceruloplas-min. From the latest EPR data, we realized that measurements of oxidation states of metals made with older techniques are no longer reliable.
The partial list in Table 2 will merely indicate the trace and bulk metals and some enzyme actions associated with them. No distinction is made here between metals which are activators and metals as integral components of metallo-enzymes.
Table 2. Enzyme And Other Functions Associated With Metals
Metals Enzymes Other Functions | ||
Mg | Acetate kinase, adenosine phosphokinaae, ATPase, choline acylase, deoxyribonuclease, dephospho-Co A-pyrophosphorylase, fructokinase, glyceric kinase, hexokinase, polynucleotide phosphorylase. | |
Ca | alpha-Amylase, aldehyde dehydrogenase (Mg), lipase | base |
V | green algae, blood of ascidian (marine worm) | |
Cr Mn | Arginase, carnosinase, prolinase, enolase, S-phosphoglvcerate kinase, aminoacidamidase, glucose-1-P-kinase, isocitricdehydrogenase, DPN (Mg). | glucose tolerance factor |
Fe | Aconitase, cytochromes, formic hydrogenylase (+Mn), homogentisic oxidase, phenylalanine hydroxylase, peroxidase, catalase. | ferritin, hemosidern hemes |
Co Ni Cu | Acetylornithinase, aspartase. | vitamin B« |
Laccase, phenolase, tyrosinase, uricase. | ceruloplasmin, cytochrome vitamin E | |
Zn Se | Acylase I (Co), carbonic anhydrase, carboxypeptidase, alcohol dehydrogenase, glutamic dehydrogenase. | |
Mo | Xanthine oxidase, nitrate reductase. | |
Continue to:


