Trace Metals And Biological Function
Description
This section is from the book "Chemistry Of Chelation In Cancer", by Arthur Furst. Also available from amazon: Chemistry Of Chelation In Cancer.
Trace Metals And Biological Function
The role of trace metals in the biological processes has by no means been clearly elucidated. Various postulates are being presented to account for the catalytic activity of some metals and unusual toxicity for others. Paradoxes always arise, for whereas minute amounts of trace metals may have one pharmacological or biochemical effect, reversals in these activities may take place if the metal concentrations rise beyond a certain limit. What takes nlaee at these limits?
The Vitamins
Metals have been associated with vitamins, proteins, enzymes, and nucleic acids. Radio iron derivatives of ascorbic acid were used to study the uptake of ascorbic acid by tumors. Results showed that tumors more closely resembled visceral organs than normal surrounding tissue.
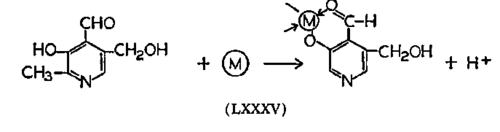
The best evidence for a vitamin-metal (LXXXV) chelation interaction is shown by the B6 group, including pyridoxal and pyridoxamine. The phenolic and the carboxaldehyde groups chelate the metal, leaving the alcoholic hydroxy free for kinase action. Another way of looking at the molecule is to assume that a hemiacetal is formed and chelation takes place between the hemiacetal and phenol (LXXXVI). Even alkali metals can be associated with pyridoxal, and here it is best to consider the quinoid form (LXXXVII).


Metal chelation of a Schiffs base formed from pyridoxal and an amino acid has been postulated as part of the mechanism of action of pyridoxal in transamination reactions (LXXXVIII).

After the formation of first the anil and then the chelate, an electron shift takes place. A proton attaches to the complex, and following a hydrolysis step, two products are formed, pyridox-amine and an a-ketoacid.
The Proteins
Metals may be associated with the plasma proteins and amino acids in biological fluids. Some transition element ions, as well as ethylenediaminetetraacetic acid, may even influence the penetration of amino acids into ascites tumor cells. That amino acids and proteins chelate readily with many metals is well documented. Binding studies of various divalent cations with glycine and simple peptides give evidence for these peptide chelates (LXXXIX). The site of chelation may involve the o-amino group.

The ability of transition metal ions to act as bridges between proteins may depend on the stability constants of the metal-protein complex. Some divalent cations may affect both size and state of protein colloids, and may also influence their solubility. When small concentrations of calcium ions are added, casein appears more soluble in water, but the addition of more calcium may force some casein out of solution. The precipitation of casein by transition element ions is temperature-dependent. It is possible to calculate the number of cations associated with one molecule of a protein like serum albumin. To relate these effects to the cancer problem would require data on how these complexes are formed under physiological conditions (6, 129,220).
Continue to:


