Equilibrium Constants, Free Energy Changes And Conformation
Description
This section is from the book "The Scientific Contributions Of The Ben May Laboratory For Cancer Research", by The University of Chicago. Also available from Amazon: The Scientific Contributions Of The Ben May Laboratory For Cancer Research.
Equilibrium Constants, Free Energy Changes And Conformation
The availability of highly purified and very active specific hydroxysteroid dehydrogenases provided the opportunity for the accurate measurements of the equilibrium constants of these reactions, and hence for the evaluation of the standard free energy changes which are involved. The total free energy change of reaction (3) may be considered as the sum of the energy changes of reactions (1) and (2):
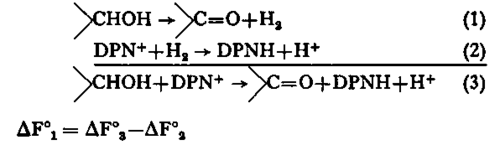
Precise measurements of ΔF°2 have given the value of +5 .22 ± 0.2 kcal. per mole at 298°K (Burton and Wilson, 1953), so that ΔF°1 may be computed from equilibrium constant measurements of the overall reaction. The equilibrium constants for the inter-conversion of a variety of steroid alcohol-ketone pairs were determined at 298° K in media of low ionic strengths and varying pH. Two equilibrium constants for pyridine nucleotide-linked reactions may be designated in accordance with the suggestion of Racker (1950):
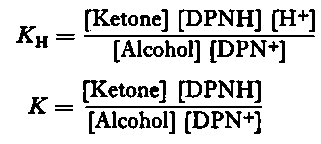
Whereas the value of KH is independent of pH, it may be seen that K is a function of the pH, such that log10 K is a linear function of unit slope when plotted against pH.
Whenever possible, the equilibrium was approached from both sides, as exemplified for the interconversion of testosterone and 4-androstene-3,17-dione by β-hydroxysteroid dehydrogenase which has been studied most intensively (Fig. 1). The validity of the measurements was examined by varying a number of experimental conditions. In separate experiments, the concentration of enzyme protein (1 to 25 µg. per ml.), the pH
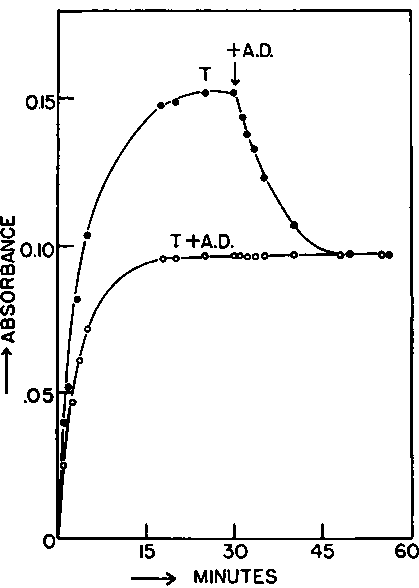
Fig. 1. Equilibrium of the interconversion of testosterone (T) and 4-androstene-3, 17-dione (A.D.) by P-hydroxysteroid dehydrogenase. Two reaction systems are shown which contained in a final volume of 3 01 ml.: 100 µmoles phosphate buffer, 0.204 nmole DPN+, 01 ml. methanol containing 0.186 µmole 4-androstene-3,17-dione and 0 169 µmole testosterone, and 216 units of P-hydroxysteroid dehydrogenase (2.9 µg. protein). The temperature was 24.8° and the final pH 7.12. In the lower graph both steroids were present initially, whereas in the upper graph the addition of 4-androstene-3, 17-dione was delayed for 30 minutes. Measurements were made at 340 mµ in silica cuvettes of 1.0 cm. light path.
(6.5 to 9.5) and the ionic strength of the buffers (0-01-0-07 M) were varied. No significant or systematic change in the magnitude of the computed equilibrium constant was detected. The mean value of 20 determinations of KH for the testosterone←→
4-androstene-3,17-dione interconversion under various conditions was 37.8 (±1-1)X10^-9M at 298°K. This corresponds to ΔF°3= +10-4 kcal./mole or ΔF°1=5.2 kcal./mole. A comparable value has been reported for the interconversion of oestradiol-17β and oestrone by a purified placental enzyme, KH= 18 (±5) x 10^-9M at 298°K (Langer and Engel, 1958). Unfortunately, no 17a-hydroxy8teroid dehydrogenase is presently available to permit studies of the oxidation equilibrium of 17a-hydroxysteroids.
Enzymic measurements of the equilibrium constants for the oxidation of several 3a- and 3β-hydroxysteroids of the 5a- and 5β-androstane series have been reported (Talalay, 19576). For these C19 compounds the equilibrium constants ranged from 6.68-7.42 x 10^-9 for the axial hydroxyl groups (ΔF°1=5-95 to 6.0 kcal./mole) and from 1 -57 to 2.11 x 10^-9M for the equatorial hydroxyl groups (ΔF°x=6.7-6.85 kcal./mole). The observed free energy differences are entirely consistent with the assigned conformations, since the equatorial hydroxyl groups are more stable (less easily oxidized). This difference may be related to the more hindered conformation of the axial hydrogens which are transferred to DPN in the rate-controlling step. Accordingly the difference in standard free energy between the axial and equatorial alcohols is about 0-8 kcal./mole at 298°K. Pitzer and Beckett (1947) calculated the energy difference between (a)-methylcyclohexane and (e)-methylcyclohexane to be approximately 2 kcal./mole. Hence, methylcyclohexane exists at room temperature almost entirely as an equatorial substituent. The observed energy differences in the steroid series are much smaller, and possibly all the members of the fused steroid ring system are not in the chair conformation. Thus, Nace and Turner (1953) have reported from dipole moment measurements that whereas 5a-androstane-3,17-dione is probably entirely in the chair conformation, 5β-androstane-3,17-dione may contain as much as 15 per cent of molecules in which ring A is in the boat conformation. In this connexion, it may also be mentioned that he effect of the angular methyl group on the energy difference between trans- and cisor-decalins is to reduce this energy difference from about 2.14 to 0.8 kcal./mole (Klyne, 1954).
Further enzymic measurements have now been made of the equilibrium constants of oxidation of the four isomeric ring A reduction products of 17,21-dihydroxy-4-pregnene-3,20-dione (Table III). The equilibrium constants (KH) for all the compounds lie in the range of 1 to 2 x 10_9m corresponding to ΔF°1= +6-7 to 7-1 kcal./mole. In these substituted 5a- and 5β-pregnane compounds the energy differences between "axial" and "equatorial" hydroxyl groups are very small. This would suggest that some of these compounds may exist in solution to a considerable extent with ring A boat conformations, which would invalidate the assignment of strictly axial and equatorial conformations.
Table III. Equilibrium Constants And Free Energy Changes In Oxidations Of C31 Steroids
Reaction | Presumptive conformation of 3-hydroxyI group | KH X 10^-9 (298° K) | ΔF°2 | ΔF°1 |
3a, 5β-THS←→5β-DHS | Equatorial | 213 | + 11.9 | +67 |
(1-94-2-36) | ||||
3p, 5a-THS←→5a-DHS | Equatorial | 1-88 | + 12.0 | +68 |
(1-68-2-16) | ||||
3a, 5a-THS←→5a-DHS | Axial | 113 | + 12.3 | +7.1 |
(1-02-1-22) | ||||
3p, 5β-THS←→5β-DHS | Axial | 0-99 | + 12.3 | +7.1 |
(0-79-1-29) | ||||
3a, 5β-THE←→5β-DHS | Equatorial | 1.07 (0.96-1.21) | + 12.3 | +7.1 |
3β-5a-THE←→5a-DHE | Equatorial | 0.92 | + 12.4 | +7.2 |
THS = 3(a or β) 17a, 21-Trihydroxy-5(a or β)- pregnan-20-one DHS = 17a, 21-Dihydroxy-5(a or P)-pregnane-3,20-dione THE = 3 (a or β), 17a, 21-Trihydroxy-5(a or β)-pregnane-11,20-dione DHE = 17a, 21-Dihydroxy-5(a or p)-pregnanc-3,ll,20-trione.
Continue to:


