Analytical Applications
Description
This section is from the book "The Scientific Contributions Of The Ben May Laboratory For Cancer Research", by The University of Chicago. Also available from Amazon: The Scientific Contributions Of The Ben May Laboratory For Cancer Research.
Analytical Applications
Enzymic resolutions of racemic compounds. Wettstein and colleagues (Vischer et al., 1956) have made ingenious use of the optical specificity of microbiological transformations for the resolution of racemic steroids. When synthetic (±)oestrone (I) was subjected to the action of fermenting yeast, (+)oestradiol-
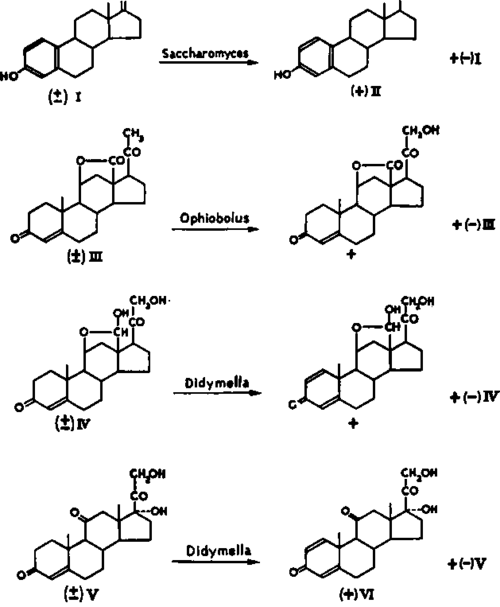
17β (II) and (-)oestrone were isolated. The microbiological resolutions of racemic steroids assumed special importance during the course of the total synthesis of aldosterone. The Swiss workers showed that only the (-f-)-isomer of the totally synthetic racemic lactone (III) underwent hydroxylation at by cultures of Ophiobolus herpotrichus, whereas the enantiomorph remained unattacked. Similarly, only the naturally occurring enantiomorph of racemic aldosterone (IV) was dehydrogenated at the 1, 2 position by Didymella ly coper ski. Under similar conditions (±)-cortisone (V) was converted to a mixture of (-)-cortisone and (+)-l-dehydrocortisone (VI). Thus, Wett-stein and colleagues have combined in a single procedure a synthetically desired reaction with a gratuitous resolution.
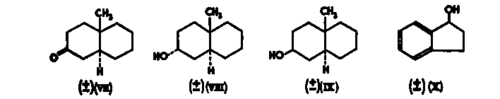
The action of p-hydroxysteroid dehydrogenase is entirely stereospecific and oxidizes only one enantiomorph of totally synthetic (±)-testosterone (Talalay, 19576). Under conditions which would be expected to lead to a complete oxidation of testosterone to 4-androstene-3,17-dione, a 96 per cent conversion was observed for 0-347 µmole of pure (+)-testosterone, whereas, under the same conditions, an equivalent quantity of (±)-testosterone* was only oxidized to the extent of 48 per cent (Fig. 2). Paper chromatography of the products of the oxidation of the racemic steroid revealed approximately equal amounts of testosterone and 4-androstene-3,17-dione, presumably representing members of the (+) and (-) series respectively (Fig. 3). Prelog and his colleagues have conducted a thorough and elegant series of investigations on the stereochemically directed microbiological reductions of various 2-ring ketones and the resolution of certain racemates, which are discussed by Professor Prelog (1959) elsewhere in this volume (p. 79). As part of a general study of the specificity of hydroxysteroid dehydrogenases, we have observed (Talalay, 19576 and unpublished) that certain saturated methyl decalols and decalones which may be considered to be analogues of steroid rings A and B, react with hydroxysteroid dehydrogenases.
* The racemic testosterone was a gift from Professor W. S. Johnson of the University of Wisconsin. The decalone was provided by Professor R. B. Woodward of Harvard University, and the decalols were gifts of Professor R. Baker of Northwestern University. The tetrahydro reduction products of compound S were prepared at the Worcester Foundation for Experimental Biology and were provided by Dr. F. Ungar.
Compound (VII) is reduced by DPNH in the presence of either 3a- or p-hydroxysteroid dehydrogenases. As stated above, compound (VIII) is oxidized by 3a-hydroxysteroid dehydrogenase and compound (IX) by p-hydroxysteroid dehydrogenase.
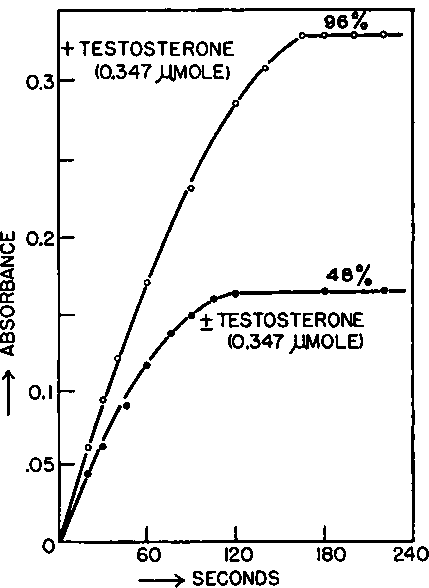
Fig. 2. Oxidation of 100 µg. (0.347 µmole) each of (+ )-testosterone and of ( ± )-testosterone by p-hydroxysteroid dehydrogenase. The reaction systems contained in a final volume of 3.0 ml.: 100 µmoles sodium pyrophosphate buffer of pH 9-5, 1.5 µmoles DPN+, 0.1 ml. methanol containing the steroids, and 1080 units P-hydroxysteroid dehydrogenase (45 ng. protein). Measurements were made at 340 mµ in silica cuvettes of 1 0 cm. light path.
(See footnote, p. 69). The rates of reaction however are very much slower than those of steroids, and the affinity constants are several orders of magnitute larger (Table I). The optical specificity of a- and p-hydroxysteroid dehydrogenases for these 2-ring compounds has not been clarified. However, (±)-indanol (X) is slowly oxidized by 3a-hydroxysteroid dehydrogenase and is not attacked by p-hydroxysteroid dehydrogenase. Only one enantiomorph of (±)-indanol is oxidized (Talalay, 19576).

Fig. 3. Paper chroma to gram of (±)-testosterone and of its oxidation product by p-hydroxysteroid dehydrogenase. Conditions of oxidation are identical with those of Fig. 2. The steroids were extracted with ethyl acetate and aliquots used for descending paper chromatography in a Zaffaroni system (1 :1 hexane-benzene as mobile phase, and 1:1 formamide-methanol for impregnation of the paper). The right- and left-hand lanes are standards: 25 µg. each of testosterone and 4-androstene-3,17-dione. The other lanes show respectively 50 µg. of ( ±)-testosterone and the oxidation products from this quantity of the racemic steroid.
The specificity of hydroxysteroid dehydrogenases for specific compounds remains only partly defined, and recently we were surprised to find that the angular methyl group is apparently essential for the reactions at C(17). In collaboration with Drs. W. L. Meyer and W. S. Johnson we subjected 13a- and 14β-isomers of (±)-18-noroestrone-3-methyl ether to enzymic reduction with p-hydroxysteroid dehydrogenase and found that neither compound was reduced by DPNH. Under the same conditions, the reduction of (+)-oestrone-3-methyl ether proceeded rapidly as expected.
Microanalysis and identification. Purified hydroxysteroid dehydrogenases have become valuable reagents for the sensitive and specific estimation of certain steroids, alcohols and ketones (Hurlock and Talalay, 1957, 1958a; Hubener and Lehmann, 1958). These procedures depend upon the quantitative enzymic oxidation of specific hydroxyl groups or reduction of ketone groups of steroids, and the spectrophotometric measurement of the accompanying stoichiometric changes in DPNH concentration. A number of considerations make enzymic methods particularly favourable for steroid microanalysis: (1) the high catalytic activity of the enzymes and their precise steric and positional specificity; (2) favourable equilibria which have permitted the development of conditions under which either quantitative oxidation or reduction of the steroid may be readily achieved; (3) the high affinities for steroids, and (4) rapid and convenient spectrophotometric measuring methods.
Procedures have been devised for the precise measurements of 3a, 3β- and 17(3-hydroxysteroid as well as 3-, 17- and 20-keto-steroids, either singly or in mixtures. These methods have been applied to the analysis of complex mixtures of steroid metabolites in human urine. The high sensitivity of these methods permits the measurement of a few millimicromoles of steroid.
It should be emphasized that the steric specificity of the hydroxysteroid dehydrogenases provides the essential basis for these methods of group analysis. These enzymic methods have also found application in the determination of the purity of steroids, the detection of contaminating stereoisomers, and have opened up the possibility for the identification and correlation of steric configurations.
Continue to:


