The Function Of Cytidine Coenzymes. Part 3
Description
This section is from the book "The Scientific Contributions Of The Ben May Laboratory For Cancer Research", by The University of Chicago. Also available from Amazon: The Scientific Contributions Of The Ben May Laboratory For Cancer Research.
The Function Of Cytidine Coenzymes. Part 3
This conclusion also offered an explanation for the experiments in which ATP, formed in situ from crystalline AMP by oxidative phosphorylation, was ineffective in supporting the conversion of P-choline to lecithin. The AMP used in these experiments was evidently free of the unidentified co-factor present in ATP lot No. 116.
A number of nucleotides were tested for the ability to replace amorphous ATP in the conversion of P-choline to lecithin. The results of one such experiment are presented in Table III. No significant activity was exhibited by ATP, ITP, UTP, or GDP. It is likely that GDP is at least partially converted to GTP in this system by the action of nucleoside di-phosphokinase (15). However, CTP was found to be much more active than the amorphous ATP. It can be seen from the data in Table III that 0.05 µmoles of CTP plus 5 pmoles of crystallized ATP is somewhat more effective in promoting the conversion of P-choline to lecithin than 5 pmoles of amorphous ATP, from which it may be concluded that the activity of the amorphous material is due to the presence of less than 1 per cent of CTP as a contaminant.
A slight activating effect is shown by CMP when tested in the presence of ATP. This is due to phosphorylation of the CMP to CTP, as evidenced by the fact that CMP is without activity when tested in the absence of ATP in experiments not shown in Table III. This finding is consistent with the observation that crystallized ATP, completely ineffective in itself, enhances the stimulatory effect of CTP (Table IV), most probably by phosphorylating CMP and CDP to CTP.
Function of Cytidine Coenzymes in Biosynthesis of Lecithin-The discovery of the requirement of CTP for conversion of P-choline to lecithin is the first demonstration of a specific r61e of a cytidine nucleotide in a major metabolic reaction. In Fig. 3 is a simplified scheme postulated to explain the r61e of cytidine nucleotides in the biosynthesis of lecithin.
The essential feature of this scheme is the intermediate formation of the novel compound cytidine diphosphate choline (Fig. 4). The phosphate monoester group of P-choline is converted to a pyrophosphate in the nucleotide CDP-choline, and is "activated" for a subsequent reaction in which the P-choline moiety is transferred to the free hydroxyl group of a D-a,β-diglyceride. The CMP which is released may then be rephosphorylated to CTP at the expense of ATP in reactions catalyzed by enzymes (15), which are known to have wide-spread occurrence. The cytidine nucleotide thus may act in a continuous catalytic fashion, carrying out a group transfer reaction during the course of which the substrate, P-choline, is built into the structure of the coenzyme itself.
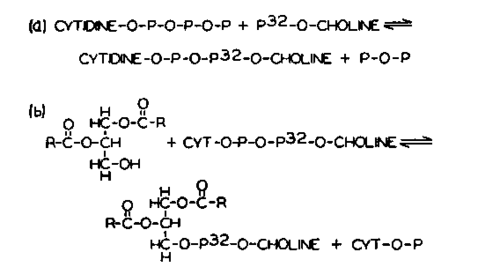
Fig. 3. The function of cytidine nucleotides in the biosynthesis of lecithin.

CYTIDINE DPHOSPHATE CHOLINE
Fig. 4. The structure of cytidine diphosphate choline.
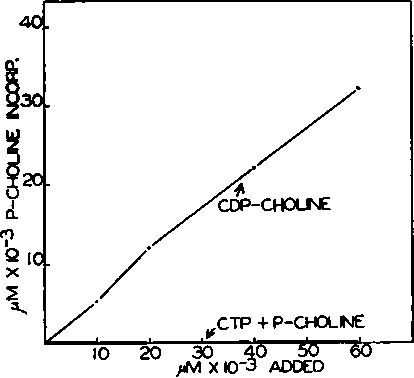
Fig. 6. The enzymatic conversion of synthetic cytidine diphosphate choline to lecithin. Each tube contained 40 pmoles of MgCl2, 100 pmoles of Tria buffer of pH 7.4, 20 pmoles of cysteine, 100 pmoles of KF, and 0.6 ml. of a suspension of rat liver particles in a final volume of 2.0 ml. Varying amounts of CDP-choline (58,000 c.p.m. per micromole) were added as shown. In the control curve, identical amounts of CTP + P-choline-l,2-C14 (58,000 c.p.m. per micromole) were added. Incubation was for 90 minutes at 37°.
Three principal lines of experimental evidence have been obtained which offer proof of the formulation shown in Fig. 3. First, highly purified synthetic CDP-choline and CDP-ethanolamine are enzymatically converted to lecithin and phosphatidylethanolamine, respectively, at rapid rates and in high yield. Secondly, CDP-choline and CDP-ethanolamine are naturally occurring nucleotides, which have been isolated from the liver of the rat and the hen, and also from yeast. Finally, enzymes have been found widely distributed in nature which catalyze the reactions shown in Fig. 3.
Enzymatic Conversion of Synthetic CDP-choline to Lecithin-The postulated scheme requires that CDP-choline should be converted to lecithin in the particles enzyme system at a rate at least equal to that of P-choline plus CTP. The availability of pure synthetic CDP-choline, prepared from P-choline-1,2-C14 by the carbodiimide method (13), made it possible to test this point directly. When labeled CDP-choline was incubated with rat liver particles, MgCl2, and buffer, the P-choline moiety was rapidly converted to lecithin (Fig. 5).
It should be noted that the yield of radioactive lecithin from labeled CDP-choline is high (50 to 60 per cent), which strongly suggests that the P-choline of CDP-choline is a direct precursor of the corresponding portion of the lecithin molecule.
The rate and extent of conversion of CDP-choline to lecithin is much greater than that observed with identical amounts of P-choline plus CTP or CMP, a finding which precludes the possibility that CDP-choline is first
Table IV. Isotope Dilution Effect Of Unlabeled Cdp-Choline On Incorporation Of P-Choline Into Lecithin
Radioactivity incorporated into lecithin, total counts | |
No CDP-choline added 1.0 µmole CDP-choline added | 645 17 |
Each tube contained 5 pmoles of P-choline-l,2-C14 (50,000 c.p.m. per micromole), 0.2 pmole of CTP, 10 pmoles of ATP (lot No. 122), 50 pmoles of Tris, pH 7.4, 30 pmoles of KF, and 0.5 ml. of a suspension of rat liver particles. The incubation was for 1 hour at 37°,hydrolyzed and the breakdown products subsequently incorporated into lecithin.
Isotope Dilution Effect of Unlabeled CDP-choline-If CDP-choline is an obligate intermediate in the reaction sequence by which labeled P-choline is converted to lecithin, then the addition of unlabeled CDP-choline to the reaction mixture should lower the specific activity of the enzymatically generated CDP-choline, and thereby depress the incorporation of radioactivity into lecithin. The experiment in Table IV indicates that the expected isotope dilution effect does occur, supporting the conclusion that CDP-choline is an obligate intermediate in the conversion of P-choline to lecithin.
Enzymatic Conversion of Synthetic CDP-ethanolamine to Phosphatidyl-ethanolamine-The reaction sequence postulated for the biosynthesis of lecithin may, in theory, be readily applied to the biosynthesis of glycero-phosphatides in general. The biosynthesis of phosphatidylethanolamine would be expected to involve the intermediary formation of cytidine diphosphate ethanolamine, a compound of structure identical with that of CDP-choline (Fig. 4), except that ethanolamine replaces the choline portion of the molecule.
Accordingly, CDP-ethanolamine was prepared from P-ethanolamine-P32 by the carbodiimide procedure (13) and tested for the ability to act as a precursor of phosphatidylethanolamine. The results of one such experiment are summarized in Table V. It can be seen that the P-ethanolamine portion of CDP-ethanolamine is converted to phosphatidylethanolamine by whole homogenates of rat liver in a yield of about 38 per cent, in a reaction analogous to the formation of lecithin from CDP-choline. When, however, use is made of a preparation of lyophilized isolated particles, which converts CDP-choline to lecithin in good yield, only a slight conversion of CDP-ethanolamine to phosphatidylethanolamine is noted. On the basis of this and other evidence, it appears probable that separate enzymes are required for the formation of lecithin and of phosphatidylethanolamine.
Continue to:


