Salol - Phenylic Ether Of Salicylic Acid
Description
This section is from the book "Dental Medicine. A Manual Of Dental Materia Medica And Therapeutics", by Ferdinand J. S. Gorgas. Also available from Amazon: Dental Medicine.
Salol - Phenylic Ether Of Salicylic Acid
Formula
C13H10O=C6H4 (OH) (CO.Oc6h5).
Derivation
Salol is obtained by the combination of salicylic acid and phenol, consisting of 60 parts of weight of salicylic acid and forty parts of phenol. It is a white, crystalline powder, insoluble in water, without odor and nearly tasteless. In the system it becomes decomposed, yielding salicylic and carbolic acids in nascent forms.
Medical Properties And Therapeutic Uses
Salol is antiseptic, germicide and antipyretic, and possesses less poisonous properties than either salicylic acid or carbolic acid alone; large doses of salol, however, cannot be administered without danger of phenol poisoning. It is accumulative, owing to its being absorbed and eliminated slowly, and hence cannot be given too frequently.
In acute and chronic renal diseases, salol is contra-indicated. It is employed internally in disorders of stomach, acute gastroenteritis, tonsilitis, gonorrhoea, skin diseases, gleet, etc. Externally used, salol is an excellent dressing for wounds, ulcers, burns, erysipelas and skin diseases. Camphorated salol is highly recommended by Cuirllier in the treatment of otitis. It can be mixed with iodoform or iodol, and the action of both be had simultaneously.
Dose
Gr. v. to ![]() www.saminda.co
www.saminda.co
Dental Uses. - Mixed with iodoform, aristol, or oxide of zinc, salol is successfully used for capping exposed pulps. It is also employed as a root-filling material by first drying out the root-canals with absolute alcohol and hot-air, then liquefying a little salol in a test-tube over a spirit lamp, and injecting some of the liquid by means of a warm hypodermic syringe to the apex of each root. It is then allowed to crystallize, when it becomes very hard.
For Erythema. Dr. A. Eichler.
Saloli..................
Zinci oxidi................
Pulv. amyli..............aa
Lanoline................
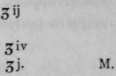
Continue to:


