Ethylated Compounds
Description
This section is from the book "Materia Medica: Pharmacology: Therapeutics Prescription Writing For Students and Practitioners", by Walter A. Bastedo. Also available from Amazon: Materia Medica: Pharmacology: Therapeutics: Prescription Writing for Students and Practitioners.
Ethylated Compounds
In experimental chemistry it has been found that the introduction of the radicle ethyl, C2H5, into an organic chemical will frequently confer upon it a sedative action. Hence many synthetic hypnotics containing ethyl groups have been placed upon the market. Ether is ethyl oxide, and common grain alcohol is ethyl alcohol. The more commonly employed ethylated hypnotics are:
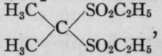
Sulfonal (sulphonmethanum), (Ch3)2C. (So2c2h5)2, a di-ethyl compound, and its tri-ethyl congener, trional (sulphonethylmethanum), Ch3.C2H5.C(So2c2h5)2, are crystalline bodies that are soluble with difficulty in water. Trional is readily soluble in whisky or alcohol. Dose, 15 grains (1 gm.). These drugs are similar in effects, but differ in their rate of absorption and their rapidity of action. Trional is the more rapidly absorbed, and being more highly ethylated, is prompter and more certain in its sedative effects. They both induce quiet and sleep without any depression of heart or medullary centers, but they do not allay pain. They have been used to check nausea, as in seasickness. They are eliminated as ethyl sulphonates, sulfonal tending to be so slowly excreted that drowsiness may persist the following day. They are usually administered in capsules, or with hot milk or whisky, sulfonal being given two or three hours, and trional one-half to one hour, before the expected sleep. The larger doses are said to be irritant to both stomach and kid- -neys. Dreams and nightmares and herpetic ulcers of the mouth are untoward effects attributed to trional.
Poisoning has occurred a number of times from their excessive use by the laity, in amounts, for example, of 200 grains (13 gm.) of sulfonal or 120 grains (8 gm.) of trional. The symptoms are chiefly gastric, renal, and mental. They are: nausea, vomiting, diarrhea, and abdominal pain, with stupor, mental confusion, hallucinations, muscular weakness, and incoordination, followed by collapse and death. Rolleston reports collapse with unconsciousness, very weak pulse, and slow breathing from 125 grains of trional. In some cases, though not in all, the urine contains hematoporphyrin from destruction of red blood-cells. It may contain albumin and casts or blood from acute nephritis, or it may be suppressed. Von Noorden has advised against the use of these drugs in nephritis because of their tendency to irritate the renal epithelium. Starr mentions them as causes of multiple neuritis. The poisoning is treated by large quantities of milk, and alkalies such as sodium bicarbonate. The alkali is to combat acidosis.
Veronal, di-ethyl malonyl urea, di-ethyl barbituric acid, C(C2H5)2.CO(Conh)2, slightly bitter and slightly soluble in water (1: 150), has an action resembling that of trional. It usually results in sleep in one-half to one hour, and this- lasts several hours, without depression of the circulation. Veronal may, however, be very slowly excreted, so that drowsiness, headache, and dizziness persist all through the following day. In some cases the sleep is dreamy, unrefreshing; and at times, particularly in old people, sleep persists for twenty-four to thirty-six hours. It is extensively employed as a hypnotic in all ordinary conditions where sleep is wanting. It is also used to some extent in epilepsy, delirium tremens, prolonged labor, and the vomiting of pregnancy and seasickness. Dose, 5 grains (0.3 gm.). A sodium compound of veronal, soluble in 5 parts of water, has been marketed under the names Medinal and Veronal-sodium. It is bitter, but may be used by rectum, or even in 10 per cent. solution, hypodermatically. Luminal is a close relative.
Toxicology
Itching of the skin, erythema and other skin rashes, conjunctivitis, and glycosuria have been reported following its use. Jacobi says that in addition to the hypnotic action it causes relaxation of the capillary walls similar to that from arsenic, with fall in blood-pressure, congestion of the abdominal viscera, and depression of respiration. It does not affect the . cardiac muscle. From 20 grains (1.3 gm.) given one night and 10 grains (0.7 gm.) the following night, the author saw a case develop a generalized enlargement of the lymph-nodes, a measles type of rash, and fever up to 103° F. which lasted a week. The diagnosis was established by the recurrence of these symptoms forthree days on two occasions following 10 grains (0.7 gm.) of medinal. The average lethal dose is 8 to 10 gm. Several deaths have been reported, as in a child of three years after 10 grains (0.7 gm.). The treatment consists of alkalies and diuresis, and that for collapse.
Bromural, monobrom-valeryl-urea, (Ch3)2CH.Chbr.Conh.-CO.Nh2., resembles veronal very closely in its effects but is less active. Dose, 15 grains (1 gm.). Isopral is a similar drug with the same dose.
Adalin, brom-di-ethyl-acetyl-carbamide, C(C2H5)2Br.Conh.-Conh2, is a substance of the same class as veronal and bromural. It is soluble freely in alcohol, but with difficulty in water, is almost tasteless, and is milder in action than veronal. Dose, 15 grains (1 gm.). A case is reported of sixty hours' unconsciousness after 45 grains (3 gm.).
Urethane, aethylis carbamas, Nh2cooc2h5, soluble in less than its own weight of water, is a mild hypnotic and diuretic in dose of 1 dram (4 gm.). It changes in the body to urea, and because of this fact is advised against in nephritis.
Hedonal is methyl-propyl-carbinol-urethane, soluble in 120 parts of water and readily in alcohol. It is incompatible with alkalies. Dose, 15 grains (1 gm.). It has been used as an intravenous anesthetic, Fedoroff (1910) reporting 330 cases. Page (1912) recommends a solution of 0.75 per cent. in normal saline infused at the rate of 50 to 150 c.c. per minute. The adult dose is 500 c.c. The respiration was deep and regular, the pulse good, the reflexes were abolished. Veale (1912) employed it in quantities up to 1200 c.c. and from the larger amounts got skin edema, pulmonary edema, bronchitis, and pneumonia, as well as thrombosis in the vein.
Amylene hydrate, dimethyl-ethyl carbinol, (Ch3)2Cohc2h5, a limpid liquid, soluble in 10 parts of water, resembles paral-dehyd in its action, but is a milder hypnotic and less disagreeable in odor and taste. Dose, 1 dram (4 c.c.) by mouth or rectum. A compound of amylene with chloral is known as "dormiol."
All the above are the hypnotics which are in common use to induce sleep where the wakefulness is not due to pain. Except chloral hydrate, which is powerful and dangerous, none of these, unless in doses above the ordinary, cause any essential depression of the heart, medullary centers, or reflexes; they are, therefore, safe general hypnotics which can be employed even in depressed states of the circulation.
Paraldehyd (Ch3coh)3 is not an ethylated compound, but may be considered here. It is a volatile liquid with a penetrating, disagreeable ethereal odor and a burning taste. It is soluble in 8 parts of water, and, freely in alcohol and the oils. Locally it resembles ether, and by its irritation of the mouth and probably also of the stomach is a reflex stimulant of the respiration and circulation. It is rapidly absorbed, and soon produces sleep without any appreciable depression of the medullary centers. The chief drawbacks to its use are its unpleasant taste, its irritant local effects, and the persistence of its odor and its taste, owing to eructations from the stomach or to its excretion in the breath. It may be administered dissolved in sweetened water, syrup of lemon, lemonade, whisky, or beer, which partly disguise the taste. It may also be given by rectum, dissolved in water. Dose, 30 minims (2 c.c.) for ordinary hypnotic effects. In the insomnia or delirium of alcoholics it is sometimes administered with excellent effect in doses of 2 drams to 1/2 ounce (8 to 15 c.c). We have seen one ounce administered in three hours with apparently nothing but good effect. Poisoning results in stupor, with depression of the medullary centers and heart muscle, and collapse. Three and one-third ounces (100 c.c.) at one dose have been recovered from. The paraldehyd habit is occasionally met with. Fonaca and Querelli (1912) record the case of a physician who took it for five years, the nightly dose reaching 1/2 ounce (15 c.c.). Then symptoms resembling those of chronic alcoholism were followed by delirium tremens with one convulsion, high temperature, weak pulse, intense sweating, polyuria, and marked thirst. Paraldehyd has been employed for intravenous anesthesia. (See page 307.)
Continue to:


