A Theory Of Metabolism
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
A Theory Of Metabolism
Rubner1 conceived that the living cell had essentially two nutritive affinities - one for fat, the other for carbohydrate. When, as in diabetes, the affinity for carbohydrate was rendered inactive, fat alone oxidized for the maintenance of the body. Rubner's theory of metabolism is given on p. 239.
The more intimate knowledge derived from the study of the action of intermediary metabolites during short periods of time compels another viewpoint. In presenting the following interpretation, the writer is keenly aware of the transitory character of all theories.2
In each mammal there is a basal metabolism. This corresponds with the minimal heat production eighteen hours after taking a mixed diet. Under these circumstances the cells are nourished by a food supply of fat and of carbohydrate, the latter supply being regulated by the liver. The basal metabolism may be acted on by food in the following ways:
(1) Amino-acid stimulation, in which some oxy- or keto-acids derived from protein metabolism stimulate the cells to a higher level of oxidative activity.
(2) Fat plethora, in which an influx of fat from the gut increases the heat production at the expense of fat itself. When fat is oxidized two carbon atoms are broken from the chain together. What form this cleavage takes is not known; it is usually pictured as productive of acetic acid. If palmitic acid broke up by successive oxidations into acetic acid radicles, one could write the following reaction:
1 Rubner: "Archiv. fur Hygiene," 1908, lxvi, 15.
2 During the discussion which followed the presentation of papers on the subject of the specific dynamic action of the food-stuffs at the International Congress of Hygiene and Demography held at Washington in 1912, Professor Rubner said: "Ich freue mich dass die Frage der 'specific dynamic action' durch neue Untersuchungen weiter gefiihrt worden ist. Die Erklarungen der Tatsachen wechseln mit der Zeit: das ist die Geschichte der Wissenschaft. Ich freue mich constatieren zu konnen, dass meine alten Untersuchungen nun endlich bestatigt worden sind".
C16H3202 + | 140 | = | 8C2H402 |
Palmitic acid. | Acetic acid. | ||
1 gram | = | 1.348 grams | |
9353 calories | = | 4706 calories |
Such a reaction involves 50 per cent, loss of heat. Perhaps the energy imparted to the cell in fat metabolism is derived from a twofold source - acetic acid and the oxidation at the -carbon atom of the fatty acid; or perhaps a substance more highly explosive than acetic acid is set free as the result of β-oxidation. In any event one may conceive of the oxidation of fat as being in the nature of successive ultra-micro explosions, which act as power for the machinery of the cells. (3) Carbohydrate plethora, in which an influx of carbohydrate from the intestine increases the heat production. When these enter the circulation alone they are oxidized to the exclusion of fat. It appears certain that the intermediary metabolites of glucose and fructose are far more readily oxidizable than fat, and on this account, when they are present, they satisfy the energy requirements of the cell and the fat is not attacked. If glucose breaks up into methyl-glyoxal and this into acetaldehyd and formic acid, the reaction would be as follows:
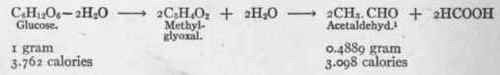
According to this computation, and assuming that hydro-lytic reactions are accomplished without thermal changes, 79 per cent, of the energy liberated in carbohydrate metabolism would be derived from acetaldehyd and 21 per cent, from formic acid.
1 If the hydrogen in formic acid were oxidized to water.
+ H2, H2 + O = H2O) the heat evolved would be 0.755 calorie. The heat of combustion of formic acid is unknown.
There is no indication of a physiologic separation of these two varieties of energy. It happens frequently that with the cessation of glucose absorption the respiratory quotient remains at 1.00, indicating that carbohydrate is still the essential food, and yet the metabolism has fallen to the basal level. One must, therefore, conclude that the metabolism increases only in the presence of a plethora of dissociated fragments of sugar. The metabolism may rise to a certain height which is not transcended, and an excess of metabolites above this level may be converted into fat (see p. 308) with scarcely any energy loss.
(4) Carbohydrate And Fat Plethora
Here there is a summation of effect. It seems as though that part of the cell mechanism which is susceptible to fat metabolism when fat is present in excess is not inhibited from metabolizing such surplus fat even in the presence of carbohydrate.
(5) Amino-Acid Stimulation And Carbohydrate And Fat Plethora
Simultaneous ingestion of an amino-acid and carbohydrate acts in such a manner as to suggest that the increase in metabolism due to carbohydrate plethora is essentially independent of that due to the chemical stimulus of amino-acids. Also when an amino-acid is given together with glucose at the height of fat metabolism (four hours after fat ingestion) the increase in heat production is nearly one amounting to a summation of the three influences.
One may, therefore, conclude that the influence of food upon the quiet resting cell under these circumstances is upon three independent mechanisms within the cell:
(a) A mechanism which is receptive to a chemical stimulus derived from the metabolism of such amino-acids as glycocoll and alanin.
(b) A mechanism of carbohydrate plethora which allows the metabolism of carbohydrate up to the limits imposed by "self-regulation".
(c) A mechanism capable of receiving power from that quota of fat which when in excess increases the heat production of the cell.
Continue to:


