Chapter XIX. Purin Metabolism - Gout
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Chapter XIX. Purin Metabolism - Gout
Uric acid was discovered in urinary calculi by Scheele in 1776, and was found to be present in gouty concretions by Wol-laston in 1797. It has since been the subject of investigations almost without number, and of theoretic speculation beyond that of any other chemical substance described in medical literature. The older work concerning the excretion of uric acid is almost valueless on account of the inadequacy of the chemical methods of the times. Accurate determinations of uric acid date from the introduction of a new method of analysis by Salkowski in 1882; and of allantoin by Wiechowski in 1908.
The newer researches are also based on more exact chemical knowledge of the precursors of uric acid. Much valuable information has been gathered as regards the normal method of production of uric acid, although it will be seen that on the pathologic side there is little beyond the conjectural to reward the student.
Emil Fischer1 grouped together uric acid, hypoxanthin, xanthin, adenin, and guanin as bodies whose varying structure depended upon slight changes around the chemical nucleus of a substance called purin. Purin, according to Fischer, may occur in the body, but on account of its ready decomposa-bility, has not been discovered there.
The relations between the purin bodies may be judged from the following formulae:
Purin....................................C5H4N4
Hypoxanthin.............................C5H4N4O
Xanthin..................................C5H4N4O2
Uric acid.................................C5H4N4O3
Adenin...................................C5H3N4NH2
Guanin...................................C5H3N4ONH2
1 Fischer: "Berichte der deutschen chemischen Gesellschaft," 1899, xxxii, 435.
Hypoxanthin, xanthin, and uric acid are respectively mono-, di-, and tri-oxypurin. Adenin is aminopurin, and guanin is aminohypoxanthin. It is evident that uric acid is the most highly oxidized product of the series, and might readily arise from the oxidation of hypoxanthin and xanthin. It is also apparent that by supplanting the NH2 group in adenin and guanin by O, they would be converted into hypoxanthin and xanthin respectively, and that from these substances uric acid might arise through oxidation.
These reactions may be thus expressed:
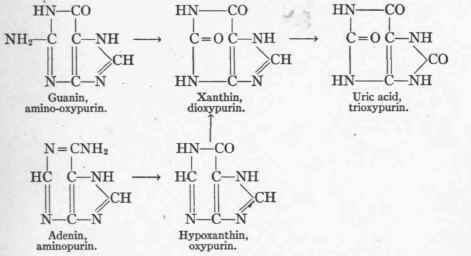
The deamination of guanin and adenin is accomplished by hydrolysis and may occur in the absence of oxygen, whereas the conversion of hypoxanthin into xanthin and the latter into uric acid are true processes of oxidation.
The knowledge of the hydrolytic cleavage products of nucleic acid is derived largely from the work of Kossel,1 who added adenin, cytosin, and thymin to chemical literature.
The formulae of the three, pyrimidin bases - uracil, cytosin, and thymin - are as follows:
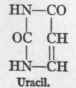
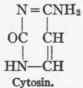
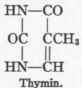
1 For the extensive literature on this subject consult the valuable monograph of Walter Jones, "Nucleic Acids," London, 1914.
Kossel and Steudel1 point out the fact that purin bases contain the pyrimidin nucleus, and that cytosin, for example, needs only cyanic acid, CONH, and an atom of oxygen to convert it into uric acid.
They query whether the pyrimidin bases are precursors or metabolized products of the purins, but the question is still unsettled.2
Mendel and Myers8 report that the pyrimidin bases, when administered intravenously or per os, reappear in the urine unchanged without increasing either the purin or the urea output. However, when nucleic acids containing pyrimidin bases are administered, the bases are not found in the urine. The pathway of their disintegration is uncertain.
Kossel's work presents the following substances as characteristic cleavage products of animal and vegetable nucleic acids:
Animal. Thymus nucleic acid: Phosphoric acid Guanin Adenin Cytosin Thymin Hexose.
Vegetable. Yeast nucleic acid: Phosphoric acid Guanin Adenin Cytosin Uracil Pentose lem of the composition of nucleic acid. They submitted guanylic acid to neutral hydrolysis under pressure, which removed phosphoric acid and left a substance (guanosin) which readily broke up on acid hydrolysis into d-ribose and guanin. These two hydrolyses may thus be written:
Bang4 extracted from the pancreas a nucleic acid containing phosphoric acid, a pentose and guanin, which compound he called "guanylic acid." The presence of a pentose in animal nucleic acid is an anomaly. Levene and Jacobs6 discovered the formula of guanylic acid, and solved the long-sought prob:
1 Kossel and Steudel: "Zeitschrift fur physiologische Chemie," 1903, xxxviii, 49.
2 Consult Abderhalden: "Lehrbuch der physiologischen Chemie," 1909, p. 381.
3 Mendel and Myers, V. C: "American Journal of Physiology," 1910, xxvi, 77.
4 Bang: "Zeitschrift fur physiologische Chemie," 1898-99, xxvi, 133.
5 Levene and Jacobs: "Ber. d. d. chem. Ges.," 1909, xlii, 2469; "Journal of Biological Chemistry," 1912, xii, 421.
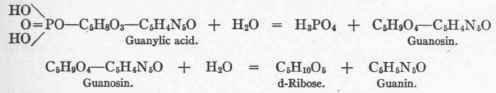
Guanylic acid is a monobasic nucleotid which yields on cleavage phosphoric acid and the nucleoside guanosin.
The more complicated nucleic acids are polymers of nucleotids. Thus, Levene and Jacobs1 give the following formula for animal nucleic acid:

Levene and Medigreceanu2 found that animal ferments have the power of hydrolytic cleavage over nucleic acid. Thus, pancreatic juice or intestinal juice through nucleinase (1) causes the resolution of a polynucleotid into mononucleotids. Animal tissues were also found to contain a similar ferment. Extracts of the mucosa of the intestine contained nucleotidase (2) which splits the nucleotids into phosphoric acid and nucleo-sids. The pyrimidin nucleosids are not further acted upon, but the purin nucleosids are broken up by nucleosidase (3) into their constituent carbohydrate and purin groups. The kidney, heart muscle, and liver contain all the above-named enzymes, and are, therefore, capable of demolishing the complex molecule of nucleic acid.
1 Levene and Jacobs: "Journal of Biological Chemistry," 1912, xii, 411. 2 Levene and Medigreceanu: Ibid., 1911, ix, 389.
Continue to:


