Purin Metabolism - Gout. Part 2
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Purin Metabolism - Gout. Part 2
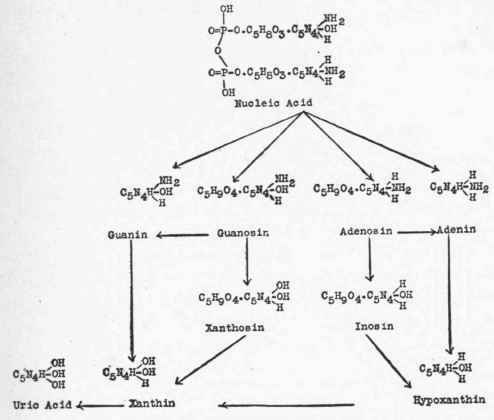
The enzyme nucleinase which breaks the polynucleotid complex of nucleic acid may not act as a simple unit. Thus, Jones and Richards1 found that when the tetranucleotid, yeast nucleic acid, was mixed with pigs' pancreas it is split into two dinucleotids; one containing the guanin and cytosin groups, the other, the adenin and uracil groups. Not only this, but Thannhauser2 found that after digesting yeast nucleic acid with human duodenal juice, the nucleotid containing uracil was split off and there remained a trinucleotid containing guanin, adenin, and cytosin. Various places of attack are therefore open.
Selecting the nucleotids forming yeast nucleic acid, one may present this summary of their transformation:

This is the simplest picture of the transformations which take place. Amberg and Jones3 have shown that the deamin-izing enzymes (see p. 531) may convert guanosin into xanthosin, which yields xanthin on hydrolysis, or convert adenosin into inosiri, which yields hypoxanthin on hydrolysis.
Jones ("Nucleic Acids," p. 77) presents the following scheme of the methods of breakdown of a guanin-adenin dinucleotid:
1 Jones and Richards: "Journal of Biological Chemistry," 1914, xvii, 71.
2 Thannhauser: "Zeitschrift fur physiologische Chemie," 1914, xci, 329; 1915, xcv, 259.
3 Amberg and Jones: Ibid., 1911, lxxiii, 407.
Horbaczewski1 was the first to note that the ingestion of nucleoproteins largely increased the uric acid in the urine. Food free from nucleoproteins has not this effect. He also found that if fresh spleen pulp, which contains no uncombined purin bases, be permitted to putrefy, xanthin and hypoxan-thin made their appearance. If now the pulp was shaken in the air, uric acid was formed from the oxidation of the bases.
Spitzer2 found that when air was passed through aqueous extracts of spleen and liver digested at 400, and with exclusion of putrefaction, uric acid was produced. The quantity of purin bases present decreased with the increased formation of uric acid. Purin bases added to such a digest were converted into uric acid, hypoxanthin, and xanthin readily and almost completely, and guanin and adenin with greater difficulty. This work established the presence of oxidizing enzymes, the xanthin oxidases, which could act on the purin bases in the organism, converting them into uric acid.
1 Horbaczewski: "Sitzungsberichte der Wiener Academie der Wissen-schaft," 1891, c, Abth. iii, p. 13.
2 Spitzer: "Pfluger's Archiv," 1899, lxxvi, 192.
Minkowski1 has shown that if a man be given hypoxanthin the quantity of uric acid increases in his urine. He also showed that if a man ingest thymus gland, the nuclein of which yields principally adenin, the amount of uric acid is increased in the urine. If the thymus be given to a dog, the uric acid plus allantoin elimination is increased. Allantoin is an oxidation product of uric acid more frequently found in dogs' than in human urine. Minkowski discovered finally that adenin when administered to a dog did not increase the uric acid elimination, and was not excreted as such, but on autopsy of the dog the uriniferous tubules were found to contain crystals the chemical structure of which showed them to be aminodi-oxypurin. In other words, adenin administered combined in nucleic acid loses its amino (NH2) group, receives three atoms of oxygen, and is thereby converted into uric acid; adenin administered as such receives two atoms of oxygen, but does not lose its NH2 group at the point for the attachment of the third atom of oxygen. This work attests a varying behavior of purin bodies in accordance with their method of chemical union with other substances, and offers a suggestive key to certain relations observed in gout (p. 546).
When theophyllin, caffein, and theobromin, the methylated purins found in tea, coffee, and cocoa, are ingested it has been stated that they are not oxidized to uric acid, but that they increase the purin bases in the urine.2 However, both Levin-thai3 and Stanley Benedict4 have found the uric acid elimination to increase in man after the ingestion of 1 to 1.5 gram of caffein daily.
The original investigations of Horbaczewski have been considerably extended by Schittenhelm and notably by Walter Jones, especially in regard to their explanation along lines of enzymotic activity.
1 Minkowski: "Archiv fur ex. Path, und Pharm.," 1898, xli, 375.
2 Kruger and Schmid: "Zeitschrift fur physiologische Chemie," 1901, xxxii, 104.
3 Levinthal: Ibid., i9i2, lxxvii, 259.
4 Benedict, S. R.: "The Harvey Lectures," 1915-16.
Jones and Partridge1 find that although the great majority of the organs of the body, when self-digested at 400 (autolysis), convert guanin and adenin into xanthin and hypoxanthin, presumably through the action of enzymes, extracts of the spleen of the pig cannot convert guanin into xanthin, although they can convert adenin into hypoxanthin. Jones therefore concludes that an enzyme, guanase, which normally removes the NH2 group and replaces it with O, is wanting in the pig's spleen, while adenase, the enzyme acting on adenin in a similar fashion, is present there. Such a reaction would read:
C5H3N4NH2 + H20 = C5H5H5O + NH3
Adenin. Hypoxanthin.
Investigating the subject further, the authors found that the pancreas contained the enzyme, guanase, which converts guanin into xanthin.
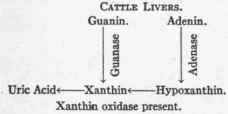
The behavior of the livers of different animals has been investigated by Jones and Austrian.2 In cattle livers, for example, adenase, guanase, and xanthin oxidase are present, whereas in dog livers guanase is present, adenase occurs in traces only, and no xanthin oxidase whatever has been found. Hence cattle livers may form uric acid from adenin and guanin, while dog livers only convert guanin into xanthin and the other processes are arrested. The process is thus graphically represented:

1 Jones and Partridge: "Zeitschrift fur physiol. Chemie," 1904, xlii, 343; see also Levene, "American Journal of Physiology," 1904, xii, 276.
2 Jones and Austrian: "Zeitschrift fur physiologische Chemie," 1906, xlviii, no.
Continue to:


