D-Alanin (Ch3.CHNH2.COOH)
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
D-Alanin (Ch3.CHNH2.COOH)
All three carbon atoms are able to enter into the formation of glucose. Found in all true proteins. In zein as much as 13.4 per cent., in muscle protein about 8 per cent, is present.
Neuberg2 found glycogen in the liver and lactic acid in the urine of a normal rabbit following the ingestion of alanin. The amino-acid had been converted into lactic acid with the elimination of ammonia. Ringer and Lusk3 gave 20 grams of i-alanin to a phlorhizinized dog and witnessed its complete , elimination in the form of urinary glucose. Dakin4 obtained the same result after administering 1-alanin.
Mandel and Lusk5 showed that d-lactic acid was completely converted into glucose in the diabetic organism and as much as 70 per cent, of the d-1-lactic acid could be transformed.
Dakin has emphasized the fact that these experiments demonstrate that the loss "of asymmetry of the central carbon atom of 1-alanin or of 1-lactic acid is essential for the formation of d-glucose. Such a loss of asymmetry would occur in the case of alanin if it were converted into pyruvic acid by oxidative deamination.
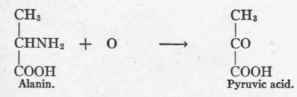
1 Neuberg: "Biochemische Zeitschrift," 1915, lxxi", 1.
2 Neuberg and Langstein: "Archiv fur Physiologie," 1903, Suppl. Bd., P. 514.
3 Ringer and Lusk: Loc. cit.
4 Dakin and Dudley: "Journal of Biological Chemistry," 1914, xvii, 451. 5 Mandel and Lusk: "American Journal of Physiology." 1906, xvi, 129.
This is a possible pathway, for pyruvic acid is convertible into glucose when administered to the glycosuric organism.1 Levene2 finds that aseptic preparations of leukocytes or kidney tissue effect no chemical change of pyruvic acid, this being contrary to the action of yeast cells which converts it into acetaldehyd with cleavage of C02 (see p. 267).
If alanin be convertible into lactic acid by hydrolysis, the asymmetry of the central carbon atom could be eliminated by a reversed internal Cannizzaro reaction, as follows:
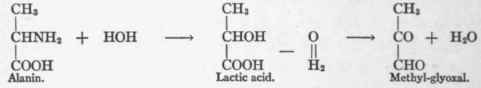
The Cannizzaro reaction involves the conversion of two molecules of aldehyd into one of acid and one of alcohol through the mediation of water. Thus, Batelli and Stern3 observed that tissue converted acetaldehyd into alcohol and acetic acid.
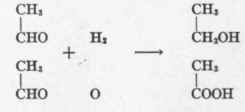
The enzyme accomplishing this reaction is called "aldehyd mutase" by Parnas.4
The internal Cannizzaro reaction deals with the oxidation of aldehyd and reduction of the keto radicles in the same compound. This may be illustrated by the conversion of methyl glyoxal into lactic acid, which Dakin5 and Neuberg6 have shown is rapidly effected by tissue in vitro. Dakin calls the enzymes accomplishing these reactions "glyoxylases," while Neuberg prefers the name "keto-aldehyd mutase".
1 Ringer: "Journal of Biological Chemistry," 1913, xv, 145; 1914, xvii, 281; Dakin and Janney: Ibid., 1913, xv, 177; Cremer: "Berliner klinische Wochen-schrift," 1913,1, 1457.
2 Levene and Meyer: "Journal of Biological Chemistry," 1914, xvii, 443.
3 Batelli and Stern: "Compt. rend. soc. biol.," 1910, lxviii, 742.
4 Pamas: "Biochemische Zeitschrift," 1910, xxviii, 274.
5 Dakin and Dudley: "Journal of Biological Chemistry," 1913, xiv, 155,423. 6 Neuberg: "Biochemische Zeitschrift," 1913, xlix, 502.
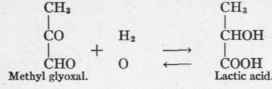
The reversed internal Cannizzaro reaction accomplishes the conversion of lactic acid into methyl-glyoxal.
Dakin1 gave 9 grams of methyl glyoxal to a phlorhizinized dog and obtained 7 grams of extra sugar in the urine, while 12 grams of 1-lactic acid yielded 9 grams of extra glucose.
These experiments enabled Dakin to picture the transformation of a d-1-alanin through d-1-lactic acid into d-glucose, as follows:
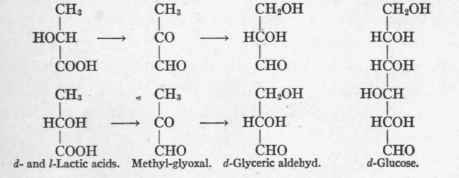
Neuberg2 reached essentially similar conclusions.
Ringer and Lusk3 showed that glyceric acid was convertible into glucose in the phlorhizinized dog, and the same was shown for glyceric aldehyd by Woodyatt.4
It has been difficult to find a chemical analogy to the transformation of the CH3 group of methyl-glyoxal into - CH2OH in glycerin aldehyd. It is certain that the CH3 groups in lactic acid and alcohol both arise in biochemical reactions from glucose, yet the manner of origin is unknown.5
1 Dakin and Dudley: "Journal of Biological Chemistry," 1913, xv, 127.
2 Neuberg: "Biochemische Zeitschrift," 1913, li, 484. 3 Ringer and Lusk: "Loc.cit.
4 Woodyatt: "Journal of Biological Chemistry," 1915, xxi, 1. 5 Neuberg and Rewald: "Biochemische Zeitschrift," 1914, lxvii, 127.
Dakin (oral statement to the writer) presents a solution of the problem dependent upon the interconversion of tautomeric forms of methyl-glyoxal:
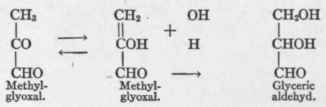
Since fructose1 and many other hexose sugars yield methyl-glyoxal with readiness in vitro, there is much likelihood that this is the intermediary substance produced when fructose and galactose, for example, are converted into glucose by the diabetic or into glycogen (which yields glucose) in the normal organism. The above described transformation, first postulated by Nef,2 is, therefore, of fundamental biologic significance not only in the metabolism of alanin but also as regards that of carbohydrate.
It may be added that alanin may be formed synthetically from pyruvic acid,3 when this substance is perfused through the liver, or from glycogen4 when an ammonium salt is perfused through a liver rich in glycogen.
It appears from this analysis that the amino-acid alanin yields on deamination an acid which may readily be converted into glucose or into methyl-glyoxal, a direct cleavage product of glucose, and which, therefore, may behave like glucose in the organism. Consideration of the oxidation of glucose will be found in Chapter IX.
Continue to:


