The Intermediary Metabolism Of Carbohydrate. Continued
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
The Intermediary Metabolism Of Carbohydrate. Continued
Levene has accomplished a large amount of work upon the intermediary metabolism of carbohydrate. He5 reported that leukocytes suspended in a Henderson phosphate mixture containing glucose induced glycolysis with the formation of d-lactic acid only, and without evidence of oxidation. This work has been confirmed by others,6 who have shown that glycolysis in the shed blood is nothing more than the conversion of glucose into lactic acid. Oppenheimer7 reports a rapid formation of d-lactic acid when d-fructose is added to a perfusing fluid and passed through a surviving liver.
Levene and Meyer8 found further that leukocytes formed lactic acid from d-glucose, d-mannose, and d-galactose, and that kidney tissue caused a formation of lactic acid from d-glucose, d-fructose, and d-mannose.
The reactions which lead to the production of d-lactic acid from the various hexoses necessitate the presence of an intermediate substance, otherwise d-1-lactic acid would frequently be the end-product.
1 Fletcher and Brown: "Journal of Physiology," 1914, xlviii, 177.
2 Parnas and Wagner: "Biochemische Zeitschrift," 1914, lxi, 387.
3 Embden, Kalberlah, and Engel: Ibid., 1912, xlv, 45; Embden, Griesbach, and Srhmitz: "Zeitschrift fur physiologische Chemie," 1914-15, xciii, 1.
4 Neuberg: "Biochemische Zeitschrift," 1913, xlix, 505.
5 Levene and Meyer: "Journal of Biological Chemistry," 1912, xi, 361; 1912, xii, 265.
6 Kraske: "Biochemische Zeitschrift," 1912, xlv, 81; Kondo: Ibid., p. 88; von Noorden, Jr.: Ibid., 94.
7 Oppenheimer: "Biochemische Zeitschrift," 1912, xlv, 30.
8 Levene and Meyer: "Journal of Biological Chemistry," 1913, xiv, 149, and ibid., xv, 65.
Wohl1 refers to the fact that methylglyoxal in alkaline solution is convertible into lactic acid. This has been shown to take place in tissue by Dakin2 and by Neuberg,3 and to be induced by white blood-cells.4
The three trioses, d- and 1-glyceric aldehyd and dioxy-acetone, yield lactic acid when treated with alkali.6 When the red blood-cells of cattle are brought into a glucose solution they have no glycolytic effect; however, they do change d-1-glyceric aldehyd and dioxyacetone into d-1-lactic acid.6 Solutions of these cells have no effect on glucose, but convert d-1-glyceric aldehyd into d-1-lactic acid. This suggests the possibility of glyceric aldehyd being an intermediate metabolite of glucose.
Embden7 has especially emphasized this method of sugar metabolism, and pictures the cleavage of glucose and fructose as follows:
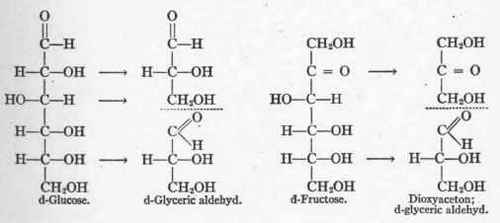
One may also conceive of the breakdown of glucose into one molecule of glyceric aldehyd and one of methylglyoxal, or into two molecules of methylglyoxal, as shown below:
1 Wohl: "Biochemische Zeitschrift," 1907, v, 45.
2 Dakin and Dudley: "Journal of Biological Chemistry " 1913, xiv, 155. 3 Neuberg: "Biochemische Zeitschrift," 1913, xlix, 502.
4 Levene and Meyer: "Journal of Biological Chemistry," 1913, xiv, 551. 5 Oppenheimer: "Biochemische Zeitschrift," 1912, xlv, 134. 6 Embden, Baldes, and Schmitz: Ibid., 1912, xlv, 108. 7 Embden, Schmitz, and Wittenberg: "Zeitschrift fur physiologische Chemie," 1913, lxxxviii, 210.

The production of methylglyoxal (CH3.CO.CHO or CH2 : COH.CHO) as an intermediary metabolite of sugar metabolism is of theoretic importance as showing by what means the asymmetry of the central carbon atom of a triose like d-l-glyeerie aldehyd may be abolished, and then through the determinative influence of living cells be transmuted into a d-compound (Dakin, see p. 193).
It will be shown later that lactic acid appears in the urine in many asphyxial conditions (see p. 422), and the long series of experiments which have been very briefly referred to above have been performed under asphyxial conditions.1 Only under these circumstances can the Cannizzaro reaction (see p. 192) take place.
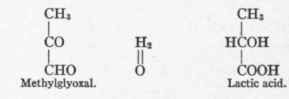
The experiments described above indicate that lactic acid is not oxidized when formed. It is, therefore, highly probable that it must first be synthesized to glucose, or at least undergo reversible conversion into methylglyoxal which is convertible into glucose (see p. 193) before it can undergo oxidation.
After consideration of all the evidence at hand, Dakin2 presents "the construction of a crude scheme aiming at the representation of the biochemical interconversions of alanin, lactic acid, methylglyoxal, glyceric aldehyd, and glucose." (Details, pp. 192-193).
1 Woodyatt: "Well's Chemical Pathology," 1914, p. 579. 2 Dakin: "XVIIth International Congress of Medicine," London, Subsection IIIa, 1913, p. 105.
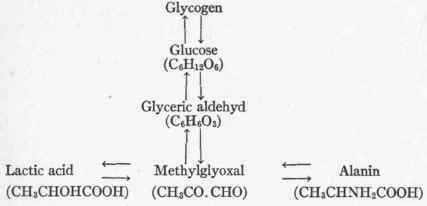
The question of the further fate of methylglyoxal in the organism is uncertain. It is known from the work of Neu-berg1 that carboxylase, which exists within the living yeast cell, splits pyruvic acid into acetaldehyd and carbon dioxid, the acetaldehyd being then either oxidized to acetic acid or reduced to alcohol.
Levene and Meyer2 find that leukocytes and kidney tissue will not cause the cleavage of pyruvic acid into acetaldehyd and carbon dioxid nor oxidize it either, so that when one comes to consider the ultimate fate of glucose in the organism the question is beset with difficulties, for the destruction of glucose depends on oxidative processes which take place within the living cell and which are not to be reproduced without the living structure. One can only formulate a hypothesis that acetaldehyd is a cleavage product of carbohydrate, as, indeed, was postulated by Magnus-Levy3 while working in Hofmeister's laboratory. From methylglyoxal, acetaldehyd might arise as follows:
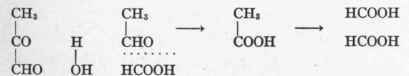
1 Neuberg: "Biochemische Zeitschrift," 1911, xxxvii, 170.
2 Levene and Meyer: "Journal of Biological Chemistry," 1914, xvii, 443.
3 Magnus-Levy: "Archiv fur Physiologie," 1902, p. 365.
In the presence of oxygen acetaldehyd would be oxidized to acetic acid, which is not convertible into glucose, and this would then be oxidized to formic acid which readily breaks up into carbon dioxid and water (see p. 302). In the presence of hydrogen, acetaldehyd is reduced to alcohol.

This, however, cannot take place to any considerable extent in the organism, for in asphyxial conditions lactic acid is the end-product of glucose breakdown, and from it alcohol cannot be evolved. The production of alcohol from carbohydrate in metabolism is thus automatically checked.
The formation of fat from carbohydrate when it is given in excess is probably due to the condensation of aldehyd molecules (Magnus-Levy). The process may thus be pictured:
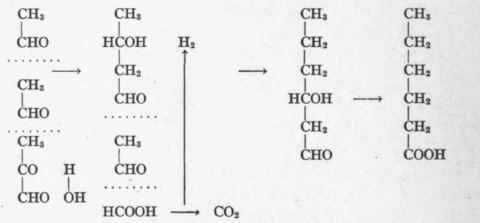
If at any time the aldehyd radicle at the end of the chain becomes oxidized, the fatty acid is completed and the process of addition terminates. In this fashion through condensation of acetaldehyd radicles split from methylglyoxal, fatty acids with even numbers of carbon atoms may be synthesized from carbohydrate in the animal organism.
Written in its simplest form the production of palmitic acid from glucose would appear as follows:
4C6H4206 = C16H32O2 + 2HCOOH + 6CO2 + 6H20
The end-result is one of carbon dioxid cleavage, just as in the case of the alcoholic fermentation of pyruvic acid induced by yeast cells.
To enter still deeper into the part played by yeast cells, and to discuss in detail the notable and fascinating work of Carl Neuberg, would carry one beyond the object of this book.1
The various sugars diffuse rapidly in the body. Thus when milk-sugar, which cannot be oxidized by the organism, is introduced intravenously into a dog, after half an hour 75 per cent, of the quantity present in the animal is found in the tissues and only 25 per cent, in the blood.2 The entrance of glucose into the cells by diffusion is accelerated by increasing their temperature.3
Continue to:


