The Intermediary Metabolism Of Carbohydrate
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
The Intermediary Metabolism Of Carbohydrate
The chemical transformations of sugar molecules present a fascinating field for the explorer. It is only possible to indicate here some of the scientific facts which are leading to a correct understanding of the subject. There is some unavoidable repetition of the facts presented in the chapter on The Intermediary Metabolism of Protein.
Lobrey de Bruyn and van Eckenstein3 found that when glucose solutions contained mere traces of hydroxyl ions, d-mannose, d-fructose, and d-pseudo-fructose appeared, and this phenomenon, called mutarotation, continued until the solution no longer rotated polarized light. Other hexoses have since been discovered in the mixture. According to Nef4 any ordinary hexose can yield 116 different substances. Of these, he was able to identify 93, of which 47 were sugars and the rest fragments of sugar cleavage. Henderson1 finds a slow but unmistakable loss of optical activity in a glucose solution maintained at the body temperature and at the alkalinity of the blood, though the quantity of sugar present is not affected.
1 Kulz: "Festschrift zu Ludwig," 1891, p. 109. 2 Jensen: "Zeitschrift fur physiologische Chemie," 1902, xxxv, 525. 3 Lobrey de Bruyn and van Eckenstein: "Receuil des travaux chimiques des Pay-Bas," 1895, xiv, 158, 203; 1899, xix, 1.
4 Nef: "Annalen der Chemie und Pharmacie," 1907, ccclvii, 214.
Nef2 suggests that the reaction d-fructose - ► glucose must take place through the intermediary formation of an enol. This may be written as follows:
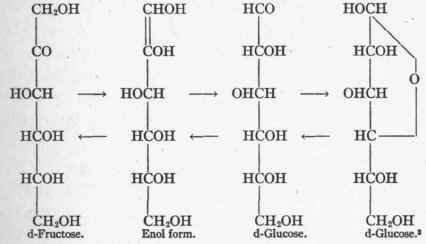
Glucose behaves like a very weak acid. In the presence of alkali, mutarotation with the production of various isomeric forms is possible. The presence of traces of acid prevents these transformations as well as any oxidative changes. To the invulnerability of the glucose molecule under these circumstances has been ascribed its non-destruction in diabetes (see p. 485). An increase in the hydrogen ion concentration of a perfusing fluid greatly reduces the utilization of glucose by an excised beating heart.4
The analogy between the phenomenon of Lobrey de Bruyn and the reactions which take place in the body is incomplete in that the latter always occur in one direction. Glucose, for example,. is not converted into fructose within the organism.
Henderson, L. J.: "Journal of Biological Chemistry," 1911, x, 3. 2 Nef: "Annalen d. Chemie und Pharmacie," 1907, ccclvii, 294. 3 Tollen's formula, accepted by Emil Fischer: "Berichte der d. chem. Gessellschaft," 1912, xlv, 461.
4 Rona and Wilenko: "Biochemische Zeitschrift," 1913-14, lix, 173.
Nef believes that the many chemical reactions of sugar may be best explained on the assumption that the sugar is in part dissociated, giving residues which may be synthesized into glucose again. Such substances might be glyceric aldehyd or methylglyoxal (see p. 193). Fragments of this sort might be open to ready oxidation in the body, or be for use as the food of yeast cells in alcoholic fermentation. When glucose or its hypothetic cleavage products, glyceric aldehyd or methylglyoxal, are treated with alkali in the presence of oxygen they are oxidized to carbon dioxid and water. If no oxygen is present, lactic acid appears in the solution. But if lactic acid be. added to an oxygenated alkaline solution of glucose, lactic acid is not destroyed. Hence, in the oxidative destruction of glucose, lactic acid in not an intermediary product.1
The relations between the trioses or sugars containing three carbon atoms and substances into which they are convertible are shown below. All of these substances when given to a phlorhizinized dog are completely converted into glucose:
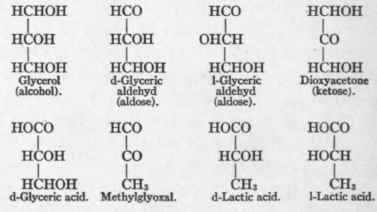
Mandel and Lusk2 gave lactic acid to a phlorhizinized dog and found it was eliminated as extra glucose in the urine (see p. 191). They furthermore found that the d-lactic acid which is eliminated in the urine of dogs poisoned with phosphorus disappears from such urine after the administration of phlorhizin. They, therefore, concluded that lactic acid arose from the metabolism of glucose, and that when glucose, its antecedent substance, was removed by phlorhizin, lactic acid vanished from the urine. They proposed the following formula of carbohydrate metabolism:
1 This description is taken from Woodyatt: "Well's Chemical Pathology," 2d ed., 1914, p. 578.
2 Mandel, A. R., and Lusk: "American Journal of Physiology," 1906. xvi, 129.
Embden1 had previously shown that d-lactic acid arises through the artificial perfusion of a liver which is rich in glycogen.
Von Furth2 has confirmed this work by demonstrating that the quantity of lactic acid eliminated in phosphorus-poisoning is increased after administering glucose. He further showed that the lactic acid elimination which occurs after cooling rabbits is increased if carbohydrate be ingested, and is prevented if the animal be freed from carbohydrate by means of adrenalin. He, therefore, concludes that lactic acid unquestionably arises from glucose.
Of similar import are the experiments of Sass,3 who showed that when strychnin convulsions were induced in dogs rendered diabetic by pancreas extirpation, though the convulsions were more powerful than in normal animals, there was no lactic acid formation.
d-Lactic acid is always found in the normal blood and muscle. Von Furth4 states that there are between 350 and 550 milligrams of lactic acid in 100 grams of fresh normal muscle of man, horse, dog, and ox.
When a muscle dies either through heat or natural rigor mortis, lactic acid is produced from carbohydrate within the muscle. Carbon dioxid is driven out at the same time, on account of the acid production.1 Parnas and Wagner2 noticed that mechanical damage to frog's muscle caused the formation of lactic acid without a decrease in the carbohydrate content. This appears to confirm the lactacidogen theory of Embden8, who found that a press juice derived from muscle formed lactic acid apparently from some unknown compound, though it left untouched added glucose, glycogen, or d-1-alanin. This substance is very likely methylglyoxal.4 Perhaps much of the lactic acid found in tissue is formed postmortem from methylglyoxal.
1 Embden: "Centralblatt fur Physiologic," 1904, xviii, 832.
2 von Furth: "Biochemische Zeitschrift," 1914, lxiv, 131; Ibid., 156.
3 Sass: "Zeitschrift fur experimentelle Pathologic und Therapie," 1914, xv, 37o.
4 von Furth: "Biochemische Zeitschrift," 1915, lxix, 199.
Continue to:


