Chapter XLIII. Purification Of Essential Oils
Description
This section is from the book "Distillation Principles And Processes", by Sydney Young. Also available from Amazon: Distillation Principles And Processes.
Chapter XLIII. Purification Of Essential Oils
An essential oil, having been " won" from the plant, is frequently in need of purification. Oils which have been produced by itinerant or native distillers can generally be vastly improved by redistillation. The colour may be bad, or the oil may have an earthy or a "burnt' odour, or, if it be old oil, it may be resinous. The usual practice is to steam-distil the oil in much the same manner that was employed to win it, but "dry " distillation may also be employed. This redistillation is generally conducted in a properly equipped distillery, where refined methods may be applied, depending on the nature of the oil.
A most important point to be observed in distilling essential oils is that, with few exceptions, they are deleteriously affected by heat. It is probably for this reason that an essential oil almost invariably has a distinctly different odour to that of the plant from which it was produced, the "cooking " to which the oil has been subjected having in some way chemicallv altered its constituents.
The constituents of essential oils, with a few exceptions, such as camphor, safrol, menthol, thymol, are relatively unstable bodies and tend to decompose or polymerise under the action of heat; thus geraniol isomerises to cyclogeraniol, anethole forms various polymers, citral decomposes to cymene and methyl heptenone, citronellal iso-merises to isopulegol, cinnamic aldehyde resinifies, and so on. Obviously therefore, it is desirable as far as possible to limit both the temperature and the duration of a distillation. The temperature can be reduced by conducting the distillation under diminished pressure. Theoretically the diminution of temperature may be so great that the boiling point of the substance could approach the absolute zero. Practically, however, we are limited by the efficiency of our vacuum pumps, the air-tightness of our stills and the efficiency of our condensers. It is a task of very great difficulty on the technical scale to obtain a diminished pressure or "vacuum " of one-fifth of a millimetre, and more often the vacuum available is of the order of 10 or 20 mm. Under a pressure of 20 mm. the majority of the constituents of essential oils boil at temperatures considerably in excess of 100° C, hence unless very efficient and refined apparatus be available it is better to resort to steam-distillation, since in this case the temperature cannot exceed 100° C.
In order still further to reduce the temperature, steam distillation under reduced pressure may be employed. This method is not frequently practised on account of two drawbacks. In the first place the amount of oil coming over with a given quantity of water is less than if a higher pressure be used,1 provided the boiling point of the oil when distilled alone is not much below that of water at the same pressure. This is shown in the following table :
Table 124
Substance. | Pressure. Mm. | Temperature. °C. | Per cent substance in distillate. |
Benzaldehyde ........................ | 76 | 45.3 | 22.5 |
Boiling point 178° C....................... | 760 | 97.9 | 31.4 |
3040 | 140.7 | 38.2 | |
Acetophenone .................................. | 76 | 45.9 | 10.4 |
Boiling point 201° C.......................... | 760 | 99.05 | 18.5 |
3040 | 142.32 | 231 | |
Camphor................................ | 76 | 46.0 | 9.82 |
Boiling point 206° C.................... | 760 | 99.08 | 21.5 |
3040 | 142.6 | 24.2 |
If, however, the boiling point at normal pressure is much below 100° C, the reverse holds.
Benzene ................................................................ | 76 mm. | 16° C. | 95.3 per ct. |
Boiling point 80° C............................ | 760 | 69.2 | 91.0 |
3040 | 112.6 | 86.9 |
Table 126 in the Appendix gives other examples.
The second drawback is the difficulty of condensing the vapour. The boiling point of water under pressure much below 50 mm. approaches the normal atmospheric temperature and there is then a strong tendency for its vapour to pass through the vacuum pumps carrying with it a proportionate quantity of the essential oil vapour. This is both wasteful and very bad for the pumps and must be carefully avoided. The boiling points of water under diminished pressures may be seen at the end of the boiling point table in the Appendix.
If the oil with which we have to deal be stable and not deleteriously affected by heat, considerable technical advantage is to be obtained by conducting the steam distillation under pressures higher than atmospheric. The theoretical aspect has already been considered.2
The use of increased pressure is of considerable value with oils having valuable constituents of very low vapour pressure such as oils of ginger, vertivert, sandalwood, cedar, and camphor, which at normal pressure only yield a low proportion of oil in a steam distillate, necessitating excessive quantities of steam for their complete treatment.
1 Cf. Theoretical section, p. 446 el seq. 2 Ibid.
As an alternative or an addition to the use of increased pressure, superheated steam may be employed with considerable advantage,1 but, as has already been pointed out, superheated or high pressure steam has a tendency to cause the hydrolysis of esters and must be used with caution if the oil contain any chemically delicate body.
"Dry" distillation has the advantage over steam or water distillation that, other things being equal, it can be conducted more rapidly, owing to the much smaller quantity of vapour to be dealt with, also, if it be desired to isolate any particular portion of the oil, this can be more accurately done by dry fractional distillation. The disadvantages are, firstly, the usually much higher temperature required ; secondly, the loss of material occasioned by vapour and residue remaining in the still, and lastly, the more costly nature of the apparatus. The first defect may largely be overcome by conducting the distillation under very low pressure ; the second, by employing steam at the end of the dry distillation to steam-distil the residue and to drive out the vapour remaining in the still.
Excepting a few instances, essential oils cannot be dry distilled under ordinary pressure without spoiling them, since the temperature required and their low conductivity for heat results in high local temperatures causing decomposition to occur, the bodies foreign to the oil produced in consequence generally possessing disagreeable odours. Such oils are said technically to be "burnt." Excessive temperature also tends to cause polymerisation or resinification, or, if the temperature be sufficiently excessive, the oil may be "cracked " and gaseous bodies produced.
There is no doubt but that the subjection of an essential oil to the action of heat acts deleteriously on its odour, and it follows that the lower the temperature at which an oil can be distilled the less is its odour damaged.
The relation between the boiling point of a substance and the pressure of its vapour is typically shown in Fig. 207, which illustrates the curve for benzaldehyde.
The relatively large reduction in boiling temperature that can be obtained by the use of very low pressure does not seem generally to be appreciated. In the instance illustrated a diminution in pressure from the atmospheric to say 20 mm. - in all a diminution of 740 mm.- lowers the boiling point by 103° C, but a further diminution of only 19 mm. causes an additional drop in the boiling point of 46°. To take another example, carvone under normal pressure boils at 230°, and under point is 22.6°. Such an extremely low pressure as this last is not practicable on a large scale, but a pressure of 1 mm. and even lower is quite within the range of modern appliances.
20 mm. at 115°, whereas at the low pressure of 0.069 mm. the boiling
1 Vide p. 447 et seq.

Fig. 207. Boiling point curve of benzaldehyde.
The duration of the distillation also has an important bearing on the odour of the distillate, and should be as short as possible. If it be desired to isolate any particular constituent of an essential oil, it is necessary to employ a fractionating column. One of the effects of a column is continually to return some of the condensed vapour to the still, where it again undergoes conversion into vapour. It follows that certain portions of the oil are subjected many times to a deleterious reflux boiling. The greater the efficiency of the column the less does this action occur, and it is a minimum with a column of absolute efficiency; that is to say, a column which allows the passage of only the lowest boiling constituent in the column and does not return any of this constituent to the still.
Columns for the separation of fractions of relatively wide-boiling range, such as terpeneless and sesquiterpeneless oils, need not be of very great efficiency, if certain oils be excepted, since the terpene and sesquiterpene constituents have boiling points differing considerably from those of the desired fraction. When, however, the terpene is present to a preponderating extent, as in the cases of lemon and orange oils, a column of high efficiency is desirable if a good yield of the terpene-less fraction is to be obtained.
Essential oils, especially those obtained by expression, frequently contain valueless bodies of very low vapour pressure, termed stearo-ptenes. These stearoptenes, if present to any considerable extent, render it difficult to obtain the last portions of the terpeneless fractions without resorting to excessive temperatures. The separation is generally best effected by distilling with steam.
Another method occasionally used to separate the terpenes from an essential oil depends on the relative insolubility of terpenes in dilute spirit compared with that of the terpeneless portions. The oil is distilled with spirit of about 40 per cent strength, using a plain still-head ; the distillate consists of a non-homogeneous mixture of dilute alcohol and essential oil, and the alcohol layer is cohobatively returned to the still, carrying with it in solution the more soluble portions of the distilled oil, while the insoluble terpene remains in the receiver. This operation is continued until the distillate coming over is homogenous, after which the alcohol in the still is distilled off and the remaining oil steam-distilled and collected separately.
A very suitable still for the preparation of terpeneless and sesquiterpeneless oils is shown in Fig. 208. The fractionating column consists of a number of tubes, through which the vapour passes, surrounded by a common water jacket p. The flow of the water through this jacket, and consequently the temperature of the column, can be regulated by means of the valve v at the bottom. A chain is suspended in each vapour tube in order to increase the surface exposed to the vapour. The still is worked under diminished pressure, and the water jacket f kept in operation until all the terpene has been distilled, after which the water is removed and the terpeneless fraction collected separately. Two receivers j and I are connected in series with the condenser G in order to facilitate the removal of the fractions without "breaking" the vacuum in the whole plant. The still is also provided with rilling and emptying valves h and d and with steam valves c and c', while the evacuation is effected through the valve k.
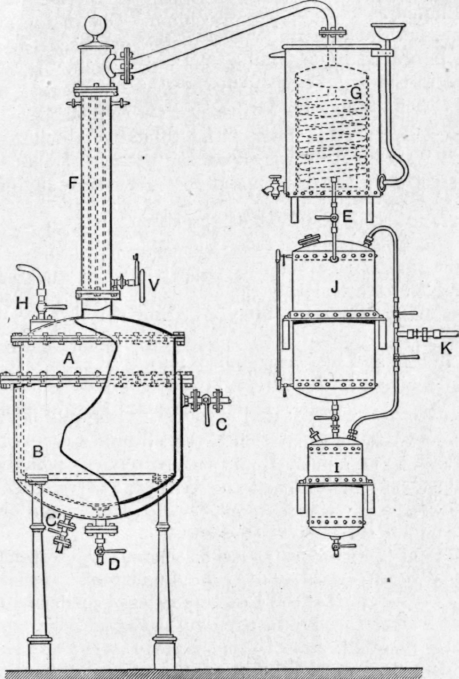
Fig. 208. - Deroy Fils AIn6, Paris.
Another type of column largely used in the industry is a modification of the Hempel column, and consists merely of a wide tube filled with broken glass or similar material. It is open to three serious objections, channeling, priming, and bad draining; but it can be easily and cheaply constructed and gives quite good results. The use of Raschig's or of Lessing's rings in the place of the broken glass tends considerably to reduce the defects.
The Condenser
The next part of the distilling plant to be considered is the condenser ; the function of the condenser is to abstract heat from the vapour, and, in consequence, to cause it to condense to a liquid state. This is not difficult to do, but a good condenser should also cool the condensed liquid so as to reduce its vapour pressure in order that loss may not occur through aerial evaporation, or if a reduced pressure is employed, in order that any important quantity of the distillate may not be lost in the vapour form through the pumps. This latter loss can occur either through inefficient condensation or on account of air leaks in the apparatus. If, as not infrequently happens with bad plant, both of these defects exist, the loss of material may be very serious.
The first demand on a condenser is, therefore, that it should be capable of extracting the heat of the vapour and of the condensed liquid very completely ; the second is that it should do so with the employment of as small a quantity of cooling water as possible.
The problem of calculating the size of a condenser is not at all an easy one as many factors are involved, some of which are of uncertain magnitude.
The only safe and most economical plan is to err on the large size. This means a bigger initial outlay, but results in economy and ease of working. We can calculate the minimum quantity of water required :
If L = Latent heat of vaporisation of the substance, S = Its specific heat in the liquid state, W = The weight of substance to be distilled, M = The weight of water required, T = The boiling point of the substance, t = The final temperature of the distillate, i.e., the temperature of the cooling water entering the condenser, T1 = The temperature of the water leaving the condenser, we have
Heat given up on condensation = WL,
Heat lost on cooling from T to t= W(T - t)S,
Heat taken up by water = M(T1 -t).
Then
M(T1-t) = WL+WS{T-t), whence or for condensing the vapour only
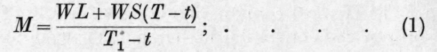
Formula (1) necessitates the supposition that the condenser is sufficiently long to allow the condensed liquid to come to the same temperature as the cooling water entering the condenser at the bottom; if this temperature be fixed, then T1 will depend on the velocity of the water, and will be equal to T as a limit.
To take an example, suppose 500 kilos of oil of turpentine to be distilled at 20 mm., when its boiling point is 53° C, we have
W = 500; L = 69; S=0-5;
T = 53° C. ; C = (say) 20° C. ; T1 = (say) 53° C. = T.
Then
(500 x 69) + (500 x 0.5 x 33) 33 = 280 gallons approximately as the minimum quantity necessary.
This is supposing the coil to be infinitely long. The rate of cooling will depend on the difference in temperature between the condensed liquid and the cooling water, neglecting the heat lag through the metal of the condenser.
A rough guide is to allow 1 square foot of cooling surface for every 1 gallon of liquid to be condensed and cooled to normal temperature per hour.
The cooling surface of a coil can be calculated from the formula
22/7 dln, where d is the diameter of the pipe, I the length of each turn, and n the number of turns.
In the case of a steam distillation, more water is required on account of the high latent heat of vaporisation of water, and the fact that, with essential oils at any rate, a large quantity of water is required to distil a relatively small quantity of oil. Thus, to steam-distil 500 kilos of limonene, which is one of the most volatile of essential oil ingredients, we require 750 kilos of water (see Appendix). To condense and cool to 20° C. these 750 gallons of water we require cooling water to the amount given by
_ (750 x 536) + (750 x 80)
80-20 ' the water leaving the condenser at 80°, whence
M = 1690 gallons.
This is required for the purpose of condensing the steam only, and a further quantity is required to condense the limonene, which can be calculated in a similar manner.
In the average worm condenser there is a dead space in the centre. This is bad, because convection currents are set up, and this tends to reduce the efficiency of the condenser on account of the cold water at the bottom mixing with the hot water from the top, resulting in increased consumption of cooling water and incomplete cooling of the condensed liquid.
This dead space should be filled up ; for instance, by a cylinder exposed on the inside to the air, or by a substance of low conductivity.
For the same reason a cylindrical containing tank is better than one of rectangular plan.
Cooling water should not be injected into the bottom of the condenser in a rapid stream, but should be broken up or baffled so as to avoid mixing with the hot water above it. Economy of water may be exercised by using aerial radiation for the first few turns of the coil before entering into the water.
In places where water is scarce, aerial radiation has to be relied on entirely; this is quite feasible, if a sufficient surface be exposed.
In a coil condenser the top coils should be of ample internal diameter in order to allow for the large volume of vapour that has to be dealt with there; the subsequent coils can diminish progressively in internal diameter. Since the ratio between the surface exposed to the cooling water and the volume of the coil is given by -, where r is the internal radius of the coil, it follows that the smaller the radius of the coil the greater the relative surface exposed.
A tall condenser is better than a broad one, since the heat gradient is not so steep, that is, the difference in temperature between adjacent coils is not so great.
Tubular condensers deal efficiently with the vapour, but the liquid resulting is apt to flow down too quickly and be inadequately cooled in consequence. This defect is sometimes overcome by raising the outlet to above the bottom of the tubes, so that they become sealed by the condensed liquid, which thus remains in contact with the cooling surface for a longer time. Where accurate fractionation is required this is bad practice.
The "Ideal" tubular condenser,1 shown in Fig. 209, overcomes this defect by retarding the flow of the hot liquid.
This condenser effects the rapid condensation and the cooling of the resulting liquid between narrow annular spaces, formed by fixing one tube inside another of slightly larger diameter, a wire being wound spirally in the space between the two tubes. The vapour to be condensed enters the annular space between the two tubes and is forced to take a spiral path by the wire ; a long path for the vapour and condensed liquid is thus provided and the cooling is effected from both sides of the annular space.
Economy of cooling water may be obtained by taking advantage of the latent heat of evaporation of water by causing the water to flow in a thin film over a chamber or coil containing the hottest vapour so that aerial evaporation of the water occurs.
1 Bennett Sons & Shears, Ltd., London.

Fig. 209. - Bennett Sons & Shears, Ltd.
The Receiver
The last item of the distilling plant needing consideration is the receiver. In the case of steam distillation the receiver should be so constructed that it will automatically separate the oil from the aqueous portion of the distillate.
Two types are necessary, according to whether the oil has a greater or a less specific gravity than water. With some oils, such as thyme, the problem is complicated by the fact that the first portions of the distillate, which contain the terpenes, are lighter than water, whereas the later portions, containing the thymol, are heavier. Thymol also has a tendency to solidify, and would choke the various pipes of the receiver; this can be overcome by allowing the condenser to run hot. Thymol melts at 51° C.
Continue to:


