Methylene Blue From Dimethylaniline. Continued
Description
This section is from the book "The Fundamental Processes Of Dye Chemistry", by Dr. Hans Eduard Fierz-David. Also available from Amazon: The Fundamental Processes of Dye Chemistry.
Methylene Blue From Dimethylaniline. Continued
Owing to its very pure shade and low price, Methylene Blue is highly valued, and is much used for dyeing tannined cotton. For silk printing the zinc-free Methylene Blue is used for the production of discharge effects. The zinc-free product is obtained by dissolving ordinary Methylene Blue in water, precipitating the zinc with sodium carbonate, and filtering off the solution of the easily soluble Methylene Blue base. By the addition of hydrochloric acid and common salt, the zinc-free Methylene Blue is precipitated out in fine crystals. On the large scale the crystallization occupies several days, arid is assisted by cooling with lead pipes through which cold water is circulated.
Of equal importance with the zinc-free salt of Methylene Blue is the nitro compound which is known as Methylene Green. The nitration is effected in a similar manner to that of Tropaeoline, and the crude zinc chloride double salt may be nitrated directly.
The crude, moist, Methylene Blue as obtained above is made into a paste with 50 c.cs. water and 20 gms. of 60 % nitric acid (400 Be.), and to it is added at 25°, 5 gms. sodium nitrite dissolved in the minimum quantity of water. The temperature is then raised cautiously to 500 with good stirring and kept there for 2 hours.
The product is diluted up with 200 gms. of saturated brine, and the precipitate filtered off after 12 hours. The crude product is dissolved in 1 litre of water at a temperature not higher than 6o°, the solution filtered and the dye precipitated by means of 150 gms. salt and 50 gms. of 50 % zinc chloride solution. After standing for 12 hours the colour is filtered off and dried at 45° until it can be powdered. It still contains about 20 % of water. If it is dried completely its strength is quickly diminished, a portion becoming insoluble. The yield from the above quantities is about 37 gms. of concentrated product.
Methylene Blue and Methylene Green are brought down to standard with dextrine, as the addition of salt diminishes the solubility too much. The most important use for Methylene Green is in combination with iron-mordanted logwood for dyeing blacks on silk, but it is also much used in conjunction with tin phosphate.
Crude Blue (2/10 mol.). 50 c.cs. H2O. 20 gms. Hno3. 5 gms. NaNo3 (approx. 93 %)• 200 gms. NaCl soln. 1 litre H2O. 150 gms. NaCl. 50 gms. ZnCl2 (50 %).
The black dyeings obtained in this manner are amongst the finest and fastest blacks for silk.
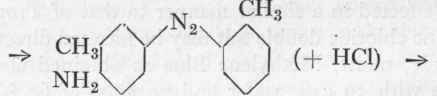
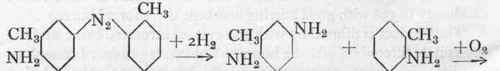
If diethylaniline or dimethyl-o-toluidine are used in place of dimethylaniline, the pure greenish Thiazine Blue, also known as Thionine Blue, etc., is obtained, which serves for the production of pure blue shades on silk; its importance, however, is decreasing owing to the competition of the faster Alizarin colours. The non-alkylated Methylene Blue, diamino-phenazthionium chloride or Lauth's Violet, is used to a very limited extent for pure violet shades. It is still made by the old method, which consists in oxidizing aniline, p-phenylene diamine, and hydrogen sulphide with ferric chloride.
Safranine from o-Toluidine and Aniline.1

o-Toluidme.
Diazoamino-o-toluidine.
Aminoazo-o-toluene,

o- Tolyl-indamine.
The methods of formation of the Azines, and of Safranine in particular, are very closely connected with those of the Thiazines (see p. 174). Of the numerous methods given in scientific literature there is only one of practical importance,2 namely, that in which
1 For more exact details, see J. Walther, "Aus der Praxis der Anilinfarben-fabrikation," pp. 21 et seq.; 291 et seq.
2 With the exception of the Anthraquinone-azines, and certain dyes of less importance.

Fig. 37. - Wrought-iron rotating autoclave with worm-drive. Weight, 11 kg. Capacity, 400 c.cs.
Working pressure, 100 atms.
Plate XV.
(as in the case of Methylene Blue), a para-diamine is oxidized with a mono-amine to give the Indamine, from which the actual azine dye is produced by the closing of the ring. In the case of Methylene Blue the thiazine ring is closed with a sulphur atom, derived from the thiosulphonic acid, whilst in the case of Safranine this position is taken up by an aromatic mono-amine, usually aniline. The mechanism of the reaction is indicated in the above scheme.
Instead of isolating the pure aminoazo-toluene, the resultant azo compound may be reduced straight away. By this means one molecule of p-diamine is formed together with two molecules of a monoamine.1 Many chemists prefer, however, to separate out the aminoazo-o-toluene first, which is then reduced, and the mixture of p-diamine with the mono-amine is oxidized to the Indamine.
On oxidizing this Indamine together with hydrogen sulphide, for example by means of ferric chloride, the homologue of Lauth's Violet is produced. As a so-called quinoid Indamine it is able to condense with a variety of substances. With aniline, under the oxidizing influence of chromic acid or recovered manganese sludge (see p. 150), a condensation product is first formed which is then oxidized to the azine by the further action of the oxidizing agent:

Piimary addition product.
o-Phenylamino-o-tolyl-indamine.

Safranine.
1 In the works, for practical reasons, enough aniline is taken to give equal molecules of aniline and o-toluidine.
In the early days of the manufacture of Safranine, an excellent oxidizing agent was made use of, namely, the recovered manganese dioxide obtained in the production of chlorine by Weldon's method. Later, as the Weldon manganese sludge disappeared from the market, use was made of chromic acid until the development of saccharine manufacture again placed large quantities of manganese sludge at the disposal of the dye manufacturers. It appears, however, that the Weldon mud cannot be altogether replaced by the manganese sludge from the manufacture of saccharine, so that at the present time the question is still unsettled as to the most advantageous method. As, however, the use of Safranine has also meanwhile diminished considerably, the question has now largely lost its importance (see also notes on Works Practice).
Continue to:


