Gas Fixtures. Burners
Description
This section is from the book "Cyclopedia Of Architecture, Carpentry, And Building", by James C. et al. Also available from Amazon: Cyclopedia Of Architecture, Carpentry And Building.
Gas Fixtures. Burners
Illuminating gas is a complex mixture of gases, of which various chemical compounds of carbon and hydrogen form the principal light-giving properties. Gas always contains more or less impurities, such as carbonic oxide, carbonic acid, ammonia, sulphureted hydrogen and bisulphides of carbon. These are partly removed by purifying processes before the gas leaves the works.
When the gas-jet is lighted, the hydrogen is consumed in the lower part of the flame, producing sufficient heat to render the minute particles of carbon incandescent. The hydrogen, in the process of combustion, combines with the oxygen from the air, forming an invisible vapor of water, while the carbon unites with the oxygen, forming carbonic acid.
Fig. 32.

Fig. 33.
Fig. 34.
Various causes tend to render combustion incomplete: there may be excessive pressure of gas, lack of air or defective burners. An excess of pressure at the burners causes a reduction of the amount of illumination; on the other hand, if the pressure is insufficient, the heat of the flame will not raise the carbon to a white heat, and the result will be a smoky flame. It therefore follows that for every burner there is a certain pressure and corresponding flow of gas which will cause the brightest illumination.
There is a great variety of burners upon the market, among which the following are the principal types:
The single-jet burner, the bat's-wing burner, the fish-tail burner, the Argand burner, the regenerative burner and the incandescent burner.
The Single-jet burner (Fig. 32) is the simplest kind, having only one small hole from which the gas issues. It is suitable only where a very small flame is required.
The Bat's-wing or slit burner (Fig. 33) has a hemispherical tip with a narrow vertical slit from which the gas spreads out in a thin, flat sheet, giving a wide and rather low flame, resembling in shape the wing of a bat, from which it is named. The common kind of slit burners are not suitable for use with globes, as the flame is likely to crack the glass.
The Union-jet or Fish-tail burner (Fig.. 34 and 35) consists of a flat tip slightly depressed or concave in the center, with two small holes drilled, as shown in Fig. 35. Two jets of equal size issue from these holes, and by impinging upon each other produce a flat flame longer and narrower in shape than the bat's-wing, and not unlike the tail of a fish. Neither of these burners require a chimney, but the flames are usually encased with glass globes.
The Argand burner (Fig. 36) consists of a hollow ring of metal connected with the gas tube, and perforated on its upper surface with a series of fine holes, from which the gas issues, forming a round flame. This burner requires a glass chimney. As an intense heat of combustion tends to increase the brilliancy of the flame, it is desirable that the burner tips shall be of a material that will cool the flame as little as possible. On this account metal tips are inferior to those made of some nonconducting material, such as lava, adamant, enamel, etc. Metal tips are also objectionable because they corrode rapidly, and thus obstruct the passage of the gas. Fig.. 37 and 38 show lava tips for bat's-wing and fish-tail burners. Burner tips should be cleaned occasionally, but care should be taken not to enlarge the slits or holes.

Fig. 35.

Fig. 36.
Fig. 37.
Fig. 38.
In all regenerative burners the high temperature due to the combustion in a gas flame is used to raise the temperature of the gas before ignition, and of the air before combustion. These powerful burners are used for lighting streets, stores, halls, etc.
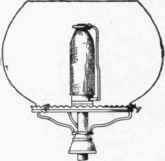
Fig. 39.
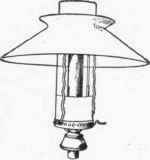
Fig. 40.
In the incandescent burner the heat of the flame is applied in raising to incandescence some foreign material, such as a basket of magnesium or platinum wires, or a funnel-shaped asbestos wick or mantel chemically treated with sulphate of zirconium and other chemical elements. A burner of this kind is shown in Fig. 39, where the mantel may be seen supported over the gas flame by a wire at the side. Fig. 40 shows another form of this burner in which a chimney and shade are used in place of a globe. Burners of this kind give a very brilliant white light when used with water gas unmixed with naphtha gases. The mantel, however, is very fragile, and is likely to lose its incandescence when exposed to an atmosphere containing much dust.
The Bunsen burner shown in Fig. 41 is a form much used for laboratory work. It burns with a bluish flame, and gives an intense heat without smoke or soot. The gas before ignition is mixed with a certain quantity of air, the proportions of gas and air being regulated by the thumbscrew at the bottom, and by screwing the outer tube up or down, thus admitting a greater or less quantity of air at the openings indicated by the arrows. This same principle is utilized in a burner for brazing, the general form of which is shown in Fig. 42. A flame of this kind will easily melt brass in the open air.

Fig. 41.
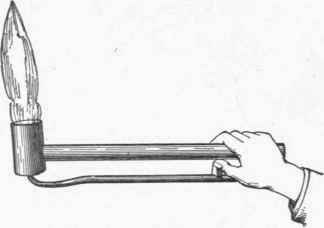
Fig. 42.
Continue to:


