Batteries. Part 8
Description
This section is from the book "American Library Edition Of Workshop Receipts", by Ernest Spon. Also available from Amazon: American Library Edition Of Workshop Receipts.
Batteries. Part 8
It is as well to mention that before turning gas on to battery, if all the cells are connected in parallel and coupled to eight 1/2-gal. Bunsens grouped in series of two for aboutl0-20 minutes,discharg-ing once or twice, but being very careful not to attack silver mesh, the intermolecular porosity of the plate will be much increased. The last charging had better remain in the plates until the gas is turned on to battery, as owing to what I call a molecular suction the freshly-liberated hydrogen from gas apparatus seems to take the place of the hydrogen disengaged in cell more readily when circuit is closed afterwards.
Now as to cost and efficiency. The cost is easily calculated, and would be between 50l. and 60l.; the absolute efficiency is difficult to calculate, but as the current density in discharge has varied from .1 to nearly .3 per sq. in. in the several kinds of plates experimented with, I think I should be justified in stating that a 25-cell battery constructed on the above lines, exposing 140sq. in., or lsq. ft. of negative area, would give about 1 electrical H.P. - a poor result from such a mighty edifice of cells - and would cost from 80l. to 90/., or not more than a first-class gas engine of the same power. Of course, future improvements would greatly increase the output of the above, and might possibly reach 2 H.P. or 1500 watts.
With regard to the working of gas batteries, there is a critical point at which the greatest effect takes place; this is that point at which the combining elements occupy the least possible space without giving or losing heat to the surrounding solution. Thus, in an oxy-hydrogen cell with perfect plates the critical point is 4° C, but the inter-molecular porosity increases with the temperature, and the temperature increases the pressure of gas at a constant volume, therefore the temperature must vary as the thermo-molecular porosity of the material, of which the plates are composed, and the pressure must vary as the temperature, to obtain a constant volume, and since it will be seen that the limit of temperature must lie between 4° C. and 100° C, in all probability unless the atmospheric pressure on positive plate is increased, the greatest effect would take place when the negative plate is heated to X00° C using a pressure of gas at 5 .21b.
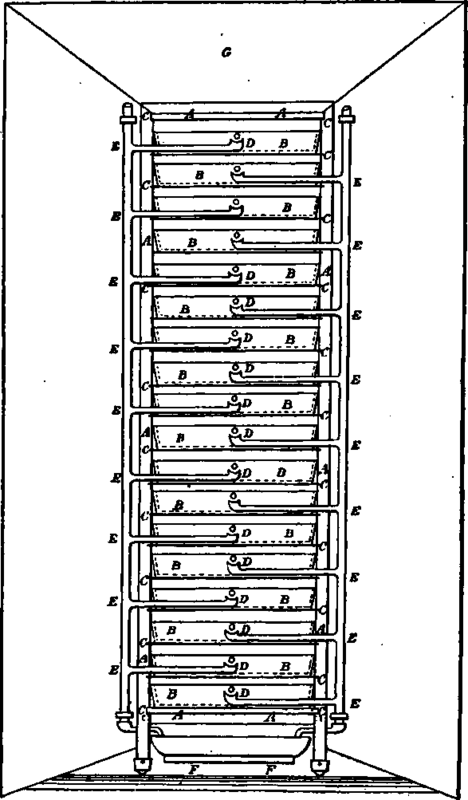
Trecby's battery.
A, vulcanised fibre frame; B, glass cells; C, plate glass supports dividing cell compartments •» D, overflow drip tubes from cell; E, catch cups and tubes to lead away overflow drip: F, basin to, hold surplus water; Q, zinc wings to concentrate draught, per sq. in., the solution remaining a 4° C.; but, of course, in actual practice, a compromise must be made, so as to give the best results. Therefore, I suggest that the negative plate be made very thin, which also increases its molecular porosity, so that the energy of the current may generate the heat required, and that the positive plate be kept cool by the evaporation which takes place, so that the solution in the immediate vicinity of the plate where the combination takes place should be as nearly as possible 4° C.; also a low temperature is more conducive to the formation of ozone in the carbon above positive plate. It may be worth mentioning that a very thin plate can be made by taking very thin metal foil and burnishing over a piece of iron wire gauze, when the metal will be pierced with tiny holes, according to the coarseness of the mesh used. (A. Treeby.)
Holtzer
The desire to obtain an open-circuit battery of large generating power, and one which should quickly recuperate, led to the construction of the Holtzer cylinder battery of the Leclanche type, shown in Fig. 85. As will be seen, the battery consists practically of two parts only, as the cylinder, zinc, and connector form the first part, they being collectively united and forming one piece alone; the second part being the glass jar. The cylinder, resting on the top of the jar, forms by its weight a well-sealed joint, thus preventing evaporation. There are no fragile parts to be easily broken, but one strong heavy carbon of cylindrical form, having a binding post well secured to it and thoroughly protected from salts. The zinc is separated from the cylinder by a porcelain piece of wedge shape to secure a close fit. Thus is presented one of the simplest and most practical forms of Leclanche' battery obtainable.

Holtzer battery.
Imchenetzki
This battery has a considerable E.M.F. and very great constancy. The exterior containing cell is of tinned iron, with a base of cast iron; the interior cell, open at the base, is of paraffined cardboard, and contains 8 porous diaphragms. Melted paraffin wax is poured at the bottom of the first cell, and when this is cold the second is introduced. There are thus 9 compartments perfectly insulated, of which 4 contain sulphate of soda with plates of zinc, and the other 5 are filled with chromic acid with electrodes of specially prepared graphite. The interior cell being a little lower than the exterior, two flattened funnels can. be placed in the spaces by which the two solutions are poured in. The liquid arrives at the same level in all the compartments. At a certain height openings are made by which the excess liquid can flow through. An outlet is made at the bottom of the first cell by which the whole of the compartments can be emptied by the simple turning of a tap. The positive electrode is in compressed graphite, with a metallic gauze, to which the terminals are soldered and covered with paraffin wax.
The graphite is light and very compact, so that the liquid in which it is immersed does not rise by capillary attraction and does not oxidise the terminals, which usually happens with other batteries. In certain types the negative pole is also made of graphite. The E.M.P. of this battery is about 2.12 volts if both electrodes are of graphite; the internal resistance is 0.08 ohms.
Iron
A novel and simple form of electric battery has recently been invented in Italy. It consists of conical vessels of cast iron and porous earthenware, with nitric and sulphuric acid. An iron cone is placed point downwards in a stand, and is partly filled with strong nitric acid. In this there is placed a cone of porous earthenware containing dilute sulphuric acid. Then follows an iron cone surmounted by an earthenware one, and so on in a series, each vessel containing its respective acid. It follows that the inner surface of each iron vessel is bathed in nitric acid, and becomes passive, acting the part of the platinum or carbon in an ordinary cell. The outer surface is attacked by the dilute sulphuric acid, and takes the place of the zinc. There are no connections to make, the simple building of the pile putting all the parts into union. The earthenware cones are 8 in. diameter and 4 in. high, and contain 550 c.c. of 10 per cent, sulphuric acid solution. The iron vessel contains 110 c.c. of nitric and sulphuric acids, the latter being 3 times the volume of the former; 60 elements, arranged in two piles, have a resistance of 10 1/2 ohms, an electro-motive force on open circuit of 81 volts, and on closed circuit of 45 volts, with a current of 4 4/10 amperes.
After 5 hours the difference of potential falls to 28 volts, and the cuirent to 2 7/10 amperes.
Continue to:


