The Analysis Of Urine. Introduction. Part 2
Description
This section is from "Scientific American Supplement". Also available from Amazon: Scientific American Reference Book.
The Analysis Of Urine. Introduction. Part 2
It is always advisable to mention the reaction to test papers of all samples received. Urine is normally acid, but there are certain diseases which render fluid neutral or alkaline. The urea of acid urine on standing is changed by a putrefactive ferment into ammonic carbonate, but this decomposition in a state of health should not take place for at least twenty-four hours. Alkalies, or organic salts of alkaline metals, when taken as medicine render the urine alkaline, and the indication is then not of much moment; but if none of these causes exist, the condition is of serious diagnostic import. Where it is desired to determine the degree of acidity of the urine voided, say, by a gouty patient, a dilute volumetric solution of caustic soda should be employed, using a few drops of an alcoholic solution of phenolphthalein as an indicator, and reporting in terms of oxalic acid. The soda solution may conveniently contain the equivalent of one milligramme of recrystallized oxalic acid (HCO.2HO) in each cubic centimeter.
Urea
Carbamide, as it is called by systematic chemists, or urea, is next to water the largest constituent of urine, and forms about one-third of its total solids. Derived from ammonic carbonate by abstracting two molecules of the elements of water, it is readily converted by putrefaction into that salt, and the urine under these circumstances becomes strongly alkaline in reaction. Earthy phosphates then fall naturally out of solution, so that the putrid fluid is always well furnished with sediment. Nitrogen that has served its purpose as muscle or other proteid leaves the animal economy chiefly in the form of urea, and its proportion in the urine, therefore, is a fair index of the activity of wasting influences.
For its determination Knop's sodic hypobromite method, on account of its convenience, is now generally preferred. The volumetric process of Liebig, which depends on the formation of an insoluble compound of urea with mercuric nitrate, possesses no advantages and is troublesome to work. The principle of the hypobromite process is simple. In a strongly alkaline solution urea is broken up by sodic hypobromite, its nitrogen being evolved in the gaseous state, and its carbon and hydrogen oxidized to carbonic anhydride and water respectively. The volume of free nitrogen obtained bears a direct ratio to the amount of urea decomposed.
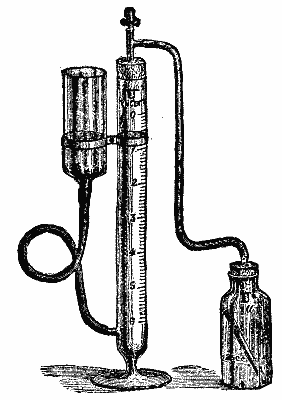
Among the number of instruments which have been introduced for the purpose of conveniently measuring the evolved gas, that of Gerrard, an illustration of which we give, is one of the simplest, cheapest, and best. The ureometer tube, b, is connected at the base with a movable reservoir, c, and by means of a rubber tube passing through a cork at the top to the generating bottle, a. To use the apparatus, fill b to zero with water and have the reservoir placed so high that it contains only an inch or so of the liquid. Replace the cork with attached tube tightly in b. Now pour into the generating bottle 25 c.c. of a solution prepared by dissolving 1 part of caustic soda in 2½ parts of distilled water, and dexterously break in the liquid a tube containing 2.2 c.c. of bromine. The tubes will be found very convenient, obviating entirely the suffocating fumes diffused in the act of measuring bromine. Allow to stand in the solution of sodic hypobromite thus prepared a test tube containing exactly 5 c.c. of the urine under examination. Cork the bottle as shown in the illustration, see that the water is at zero, and that the liquid in the reservoir is at the same level, and then allow the urine to gradually mix with the hypobromite solution. Cool the evolved gas by placing the bottle in cold water, adjust the levels of the water in the tube and reservoir (to obviate a correction for pressure), and read off the percentage of urea in terms of which the tube is graduated. Stale urine, the urea of which has largely been converted into ammonic carbonate, still yields a very fair result, that salt being also completely split up by the powerful oxidant employed.
Should the urine contain albumen, it is advisable to remove it by boiling and filtering, as, although only slowly decomposed by the hypobromite solution, it communicates to the liquid such a tendency to froth that the disengagement of the nitrogen is seriously impeded. Most of those alkaloids which might possibly be present do not yield the gas when treated in this manner, and therefore may be disregarded.
Sugar
Glucose, so characteristic of diabetes mellitus, is not difficult of detection or estimation. The facility with which it reduces alkaline cupric, argentic, bismuthous, ferric, mercuric salts, indigo and potassic picrate and chromate solutions has been utilized for the preparation of several ready methods for its determination. Trommer's test consists in adding enough cupric sulphate to color green, then excess of alkali, and boiling. Yellow to brick-red cuprous oxide forms as a heavy precipitate if glucose is present. The organic matter of the urine prevents the precipitation of cupric hydrate on the addition of the alkali. This test is delicate and deservedly popular. Fehling's well-known solution contains sodio-potassic tartrate, which serves the purpose chiefly of retaining the copper in solution. Unfortunately, Fehling's original solution has a tendency to become hyper-sensitive if kept long, a proneness to change that is much increased on dilution. When so altered, the solution will yield a more or less copious precipitate of cuprous oxide on merely boiling, and quite independent of the presence of glucose. This decomposition is obviated by preserving the copper salt in a separate solution from the tartrate and alkali, and mixing before use.
Schmiedeberg substitutes mannite and Cresswell glycerin for the Rochelle salt, in order to render the solution stable. Some prepared by the writer over twelve months ago, according to the suggestion of the latter physician, has since shown no signs of decomposition, and is now as good as it was then. For qualitative purposes the solution may be prepared thus: Dissolve 35 gm. of recrystallized cupric sulphate and 200 c.c. of pure glycerin in 100 c.c. of distilled water. Dissolve separately 80 gm. of caustic soda in 400 c.c. of water. Mix the solutions and boil for a quarter of an hour. A small amount of reduction from impurity in the glycerin takes place. Allow to stand till clear, decant, and dilute to 1,250 c.c. Ten cubic centimeters will then equal roughly 5 centigrammes of glucose. For exact quantitative determination it is necessary to standardize the solution with pure anhydrous dextrose.
Continue to:


