Estimation Of Ethyl Alcohol In Fusel Oil
Description
This section is from the book "Alcohol, Its Production, Properties, Chemistry, And Industrial Applications", by Charles Simmonds. Also available from Amazon: Alcohol: Its Production, Properties, Chemistry, And Industrial Applications.
Estimation Of Ethyl Alcohol In Fusel Oil
This is of some importance, since fusel oil containing not more than 15 per cent. of " proof spirit " is allowed to be imported into this country free of duty. As a complement to this, such fusel oil is allowed to be sent out from home distilleries also without payment of duty. The accurate estimation of ethyl alcohol in such a complex mixture of alcohols as fusel oil is a matter of some difficulty, since the complete elimination of propyl and isobutyl alcohols is impracticable. The following method, however, due to J. Holmes, gives results which are substantially correct, and it may be used with confidence for fusel oils containing up to 20 per cent. of proof spirit. Though somewhat tedious in the detailed description, the actual operation takes only about two hours, or less if a series is being done.
D D
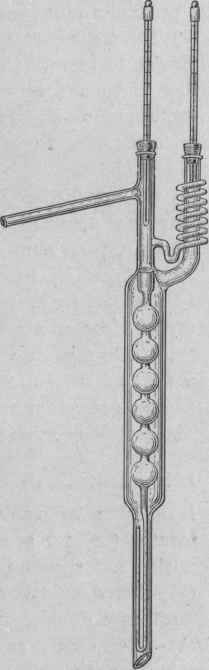
Fig. 39. - fractionating column for fusel oil analysis.
Before giving the modus operandi it will be well to explain shortly the principles on which the process is based.
(1) The oil is extracted with a relatively large volume of water, sufficient to remove the whole of the ethyl alcohol. Some of the higher alcohols are necessarily taken out at the same time. On saturating the aqueous solution with common salt and extracting it with petroleum ether, the greater part of these higher alcohols is removed.
(2) On now distilling the spirituous salt solution, all the remaining alcohols are obtained in a relatively small volume, and a repetition of the salt-petroleum extraction removes all but a small quantity of the higher alcohols, leaving the ethyl alcohol approximately pure, but not entirely so.
(3) The disturbing effect which the small quantity of higher alcohols still present exerts upon the specific gravity of the solution can now be determined by means of the refractometer, and the necessary correction applied.
The modus operandi is as follows: -
I. Extraction.
(1)
Take 75 c.c. of the sample. Shake it vigorously in a separator {A) for some minutes with 150 c.c. of water. (If an emulsion forms, add a few crystals of salt.)
(2)
Draw off the aqueous layer into a second separator (B), saturate it with salt, and extract it with 150 c.c. of petroleum ether.
(3)
Run off the aqueous saline layer into a third separator (C), and extract it with a second quantity (150 c.c.) of petroleum ether. Finally draw off this saline layer into a distilling flask. Leave the two quantities of petroleum ether in the respective separators B and G.
(4)
Meanwhile, the oil in separator A is shaken a second time with 150 c.c. of water. The aqueous extract is run into the petroleum ether remaining in separator B, saturated with salt, and extracted with this petroleum ether. Run the saline liquid into the petroleum ether remaining in separator C, extract it with this ether, and finally add it to the first quantity in the distilling flask.
(5)
Repeat (4). Thus the sample has been extracted with three separate quantities of water, and the aqueous extracts, after addition of salt, have each been extracted twice with the same two quantities of 150 c.c. petroleum ether.
Continue to:


