Aromatic Amines
Description
This section is from the book "Chemistry Of Chelation In Cancer", by Arthur Furst. Also available from amazon: Chemistry Of Chelation In Cancer.
Aromatic Amines
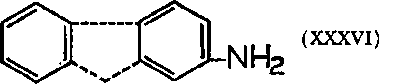
Two aromatic rings are not essential for carcinogenicity, but it is doubtful if unsubstituted aniline is a carcinogen, at least for rabbits and guinea pigs. A derivative, however, o-toluidine, has been reported active. Early interest focused on 2-naphthylamine which on ingestion was found a bladder carcinogen for both man and dogs. Other species also respond: when 2-naphthylamine is mixed with paraffin wax and implanted in the lumen of the bladder of mice, definite papillomas occur. In rats this technique is not as effective.
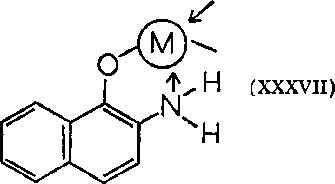
After a study of the metabolism of 2-naphthylamine, a suggestion was made that one of the possible metabolic products must be the true carcinogen. This proved to be the chelating agent, I-hydroxy-2-aminonaphthalene (XXXVII)
. Ordinarily, in the metabolism of any aromatic compound an ortho-hydroxy derivative is formed which is usually excreted as the conjugated sulfate or glucuronide. Studies were also made using radioactive 8-C14-naphthylamine to see how rapidly absorption does take place. Other conjugates and metabolic products are also known; these include products with ortho dihydroxylation on the ring that does not contain the amine.
Additional factors, such as the nature of the conjugate of the metabolic product, may be involved in the carcinogenicity of this molecule. It is possible that the phosphate, sulfate or glucuronide derivative may be important; glucuronide formation favors carcinogenesis. This implies that species with high glucuronadase activity in the bladder will be more susceptible to these carcinogens. However, the primary fact remains that in those species which metabolize 2-naphthylamine into a chelating agent, this compound is carcinogenic. This is especially true when a comparison is made for man, dog, cat, rabbit and mouse.
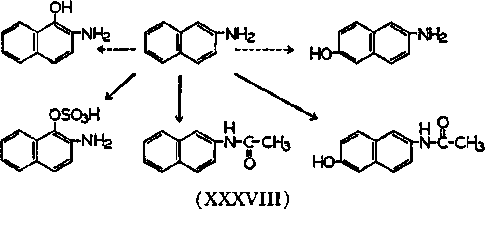
The scheme for metabolism may be shown as in XXXVIII. The amount of 1-OH compound formed and tumor production can be correlated. Dogs and man form a high percentage of the chelating agent and are quite sensitive to this compound; rats, cats, mice and rabbits form only a small amount of the chelating agent, and are affected much less. In support of the chelate hypothesis data are available to show that an isomer of the metabolic product of 2-naphthylamine, namely the 1-amino-2-hydroxynaphthalene, which is also a chelating agent, is active too. A similar derivative, 1-phenylazo-2-hydroxynaphthalene, is a chelating agent and is also carcinogenic. This last compound may possibly be reduced in the body to the 1-amino-2-hydroxynaphthalene. The converse may also be true, and some methoxy aromatic amines which are not chelating agents are also not carcinogenic. Not enough attention has been paid to the fact that the chelators in the naphthalene series are also angular. Substitutions in the 1,2- positions impart some geometric properties that resemble the beginnings of a phenanthrene structure. See nucleic acid and hydrocarbon interaction in speculation section.
Other aromatic amine carcinogens are metabolically hydroxy-lated in the ortho-position. Xenylamine (4-aminobiphenyl) has recently been reported carcinogenic in man; earlier it was found carcinogenic in animals, as was the 3,2'-dimethyl derivative (572, 574). An investigation of the metabolism of the amino-biphenyls showed that the ortho-hydroxy chelating derivatives were formed. The dog converted the xenylamine into the inactive 4-aminobiphenyl-3-sulfate; this compound, however, was easily hydrolyzed into the 3-hydroxy-4-aminobiphenyl, a more active carcinogen than the original. The 3,2'-dimethyl-4-aminobiphenyl, another carcinogen of this class, may also be metabolized to an ortfco-hydroxy derivative. Rats were able to reduce in vivo 4-nitrobiphenyl into the active carcinogen xenylamine; this was also true for dogs.
Benzidine, also believed to be an occupational carcinogen for the human, is not active in all animals. Again, it was found that humans metabolized benzidine into the chelating agent, the ortho-hydroxy derivative. In this case the 3,3-dihydroxybenzidine was formed. Metabolic products, isolated from the urine of workers in a benzidine factory, were used to induce tumors in mice.
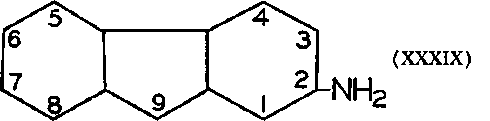
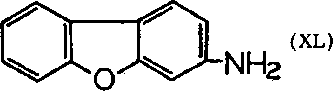
During a toxicological investigation of the potential insecticide, the acetyl derivative of 2-aminofluorene (XXXIX) (AAF) was found carcinogenic in rats, and similar to benzidine in activity on sebaceous glands, colon, liver and bladder. The metabolic studies of this agent showed that deacetylation was a primary step. At first the only hydroxylated products isolated were 7-hydroxy or the 5-hydroxy derivatives; later, however, the ortho-hydroxy compounds were found (438, 583,588); it is of special interest to note that the 1-hydroxy as well as 3-hydroxy derivatives have been isolated. Guinea pigs metabolize AAF exclusively to the 7-hydroxy compound, and AAF is non-carcinogenic for the guinea pig. A recent summary of these compounds appeared. Variations on the structure of 2-aminofluorene, including the N-methyl derivatives, are also being investigated. It will be interesting to see if the metabolic products of these carcinogens are ortho-hydroxy compounds, and hence chelating agents. Since one of the isomers of 2-acetylaminofluorene, namely 3-acetylaminofluorene, metabolizes into the chelating agent, 2-hydroxy-3-acetylaminofluorene, the eventual biological testing of this compound also should be interesting. The analog of aminofluorene, 2-aminodiphenyleneoxide (XL), has also been found to be carcinogenic.
Parenthetically, the analog, the 2-aminocarbazole, should also be tested in rodents to see if hepatomas or bladder carcinomas develop. More complex carbazoles, including dibenzcarbazoles, were found active: 1,2,5,6- and 3,4,5,6-dibenzcarbazoles (XLIA)
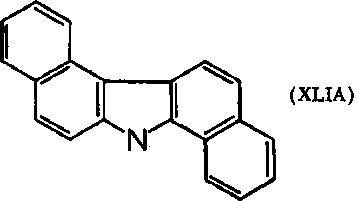
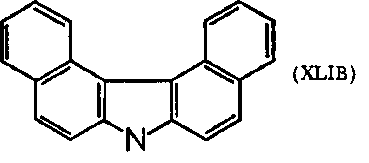
(XLIB) gave skin tumors on mice when painting techniques were employed; the latter compounds gave hepatomas in strain A, CH, and C mice. Females were more susceptible than males. Note that these two dibenzcarbazole compounds can be considered nitrogen analogs of the carcinogenic hydrocarbons.
Among the other aromatic amine compounds tested, aniline, as stated previously, is in the controversial category. Its derivative o-aminophenol, is a chelating agent, but probably not carcinogenic. It should be noted that o-phenylenediamine, a compound structurally similar, is an anti-cancer agent.
That all carcinogenic aromatic amines metabolize into ortho-hydroxy compounds, and hence chelating agents, seems to be supported by a plethora of examples. Also, the notion that chelation may be involved in carcinogenesis has been considered. The implication is that any aromatic amine which metabolizes into an ortho-hydroxy derivative is a potential carcinogen. How true is this? The metabolic pathway for the sulfa drugs is via the 3-hydroxy derivative, a chelating agent. It is therefore not surprising that sulfamethazine, sulfathiazole, sulfamethylizole, and sulfapyridine when tested were able to produce tumors in rodents.
To strengthen the hypothesis, the well-known chelating agent, 8-hydroxyquinoline, was tested as a carcinogen and proved to be positive. This may be important, since this compound was used in many contraceptive preparations. Since chelating aromatic amines are all considered potential carcinogens, predictions can now be made by referring only to the structure of the nitrogen compound. For example, it is probable that 2,3-dipyridylquinoxaline is a chelating agent and would therefore be carcinogenic. Recently it was found to be so.
Reasons for carcinogenic activity of some aromatic amines may include, in addition to the ortho-hydroxylation mechanism, an alternative explanation. These carcinogens may modify metabolism of a normal body constituent by inhibiting a catabolic enzyme so as to permit the accumulation of a chelating compound. In tryptophan metabolism of animals with bladder cancer (67, 68), a biosynthetic product is 3-hydroxyanthranilic acid, a carcinogen produced by endogenous metabolism. This compound was able to produce leukemia in RFH mice. Addition of either indole or tryptophan to the diet of animals being fed acetylaminofluorene enhances the tumor incidence. In bladder cancer patients, however, only half of those tested had abnormal tryptophan metabolism. The metabolic pathway of tryptophan is to be seen in XLII. Carcinogens are blocked out.
The ort/io-hydroxy amine, 3-hydroxyanthranilic acid, is accumulated in the animal body when 2-acetylaminofluorene is fed. In turn, this tryptophan metabolic product is further metabolized to either an ortho quinone of benzoic acid, again a chelating agent, or to quinolinic acid. The enzymes which metabolize the hydroxy anthranilic acid are blocked by acetaminofluorene, an inhibition that is reversed by DPN. Evidence has been presented that aromatic amines do mediate the oxidation of reduced pyridine nucleotides by peroxidase in the presence of manganous ion.
Continue to:


