The Enzyme Cancer Problem
Description
This section is from the book "Chemistry Of Chelation In Cancer", by Arthur Furst. Also available from amazon: Chemistry Of Chelation In Cancer.
The Enzyme Cancer Problem
Data from model metal-dependent enzyme systems may shed light on the metal-cancer problem. Since metals can displace one another in enzyme systems, there are a variety of ways in which the action of abnormal metals may be manifested and thus lead to abnormal reactions or unusual kinetic actions. Cancer tissue may contain only normal enzymes which act at different rates, yet which, on isolation, exhibit only normal behavior. Suggestions that trace metals act as bridges between enzymes and substrates imply that an abnormal metal with a similar electronic configuration may also be a bridge. If one tetrahedron ion replaces another with the same geometry, the specificity of the enzyme may change, and abnormal substrates may be bound. Even if the essential metal is complexed to the appropriate enzyme, an exogenous dye with functional groups of the proper spacing can also bind the metal. This can lead to a protein-metal-dye complex (XCV) as suggested in the literature. The normal substrate may not now be able to form an active complex with the enzyme, if by mass action the dye displaces that substrate and occupies the binding site.
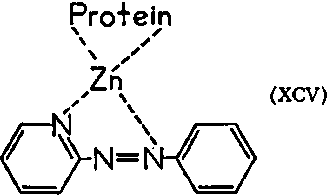
Another possibility is that an abnormal substrate (S'), which could not combine with the enzyme (E) and normal metal (M) to form an E-M-S' complex, may first chelate a foreign metal (M'), and then in this new combination replace the normal metal (M) and now form an E-M'-S'. These actions may, to a very large extent, depend on the polar nature of the side chain on the protein enzyme and also on the pH for optimum activity of the enzyme. The pKa values of these polar binding groups vary from strong acids to strong bases. Examples are 2 or 7 for phosphate, 3 for carboxyl, 5.6 for histidine, 6.5 for ammonium ion, 8.3 for sulfhydryl, 9.8 for phenolic, 11.6 for guani-dinium and greater than 14 for alcoholic or amide groups. The neighboring amino acid in the sequence may also be important in metal-binding. More work needs to be done on these relationships.
A chelate may simply bind to the metal of a normal E-M-S complex and thereby inactivate it. This may account for some of the inhibitory action of oxine (XCVI).
Cations, and especially abnormal metals, may inhibit enzyme systems by combining with coenzymes like FAD or DPN and, for all practical purposes, withdraw them from activity (38, 228).
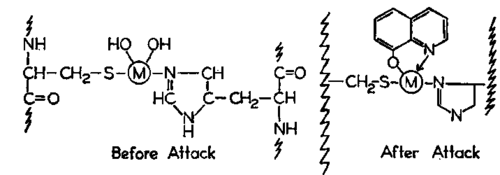
(XCVI)
Abnormal metals may act as enzyme inhibitors by ion antagonism; they may simply displace the metal activator from the enzyme. Examples of divalent cations which are antagonists to enzyme systems are given in Table 3.
Table 3. Enzyme-Inhibiting Divalent Cations
Enzyme | Normal Activator | Abnormal Metallic Inhibitors |
Carbonic anhydrase Tyrosinase Leucine aminopeptidase Xanthine oxidase | Zn Cu Mn Mo | Ag, Au, Hg, V, Cd Ag, Au, Hg Cd, Hg, Pb Hg, Ag, Pb, As |
Another conclusion possible in regard to apoenzyme or co-enzyme-metal-substrate, at least in some systems, is that different enzymatic functions of one coenzyme may require different metals. As an example, we may return to the chelating action of the pyridoxal amino acid anils (LXXXVIII). To date, no special element has been found to be the essential co-factor for these systems. Pyridoxal is essential for a variety of enzymes, especially those concerned with decarboxylations of amino acids, the transport of amino acids into cells, certain transamination reactions, the metabolism of some sulfur-containing compounds, and in the biosynthesis of niacin from its precursor tryptophan. Because each one of these biochemical pathways requires a different amino acid for anil formation with pyridoxal prior to chelation, it seems logical that no one metal could satisfy all of the conditions for these different systems. It may be that copper would be essential for one reaction, manganese for another, and zinc for a third. Thus, one should look at different enzyme systems, using methods which can detect individual metals. Perhaps activation analysis should be used to identify the metal and then a paramagnetic resonance (EPR) study would be in order for a study of the oxidation state of the cation during these enzyme reactions. These data would determine if a metal is actually essential for the enzyme activity.
Enzyme kinetics may be modified by an abnormal metal. In studies where the cations Zn, Cu, Mg, Mn or Co are enzyme activators, Hg, Ag or Au may act as inhibitors. In excess, even normal metals can also be inhibitors for other normal metal-dependent enzymes: copper will antagonize zinc in carbonic anhydrase; zinc will inactivate cobalt in glycyl-glycine peptidase; iron will antagonize copper in tyrosinase; the activity of tyrosinase and hence the amount of pigmentation in melanin is variable depending on the concentration of an abnormal metal. In very low concentrations the abnormal metal can inhibit this enzyme; in higher concentrations it can stimulate it. Similar activity is noted in the effect of various metals on monoamine oxidase. The kinetics of these enzymes can be modified as noted, in both directions. By completely displacing an essential element, or by presumably combining with a sulfhydryl group of the protein, an abnormal metal can even completely inhibit an enzyme reaction. A postulate may be made that if an abnormal metal equilibrates with but does not completely displace a normal metal in an enzyme system, the activity may be stimulated. For an example, one can return to the monoamine oxidase system and study the effect of addition of unusual cations. When either a millimole of Mg (+2) or Ti (+4) is added, no change in kinetics is noted. On addition of Ti (+3), V (+3), V (+4) or Co (+2), stimulation is observed; on the other hand, inhibitory action can be found when either Mn (+2), Cd (+2) or Hg (+2) is made part of the system.
Because both stimulatory and inhibitory effects are possible with abnormal metals, it is possible that their presence may only alter quantitatively the rate of activity. These may be nonspecific reactions; for since zinc, cobalt, nickel and manganese can sometimes substitute for one another, besides modifying the speed of the reaction, abnormal metals may also act to displace the equilibrium of an enzyme-catalyzed reaction.
Applying these observations to the cancer problem, one of two possible cases can be postulated. An equilibrium between enzyme, normal metal, chelating agent, and abnormal metal may result in a cell synthesis rate which exceeds the ability of the unaltered catabolic enzymes to degrade this new tissue. This can be diagramed in Figure 2.
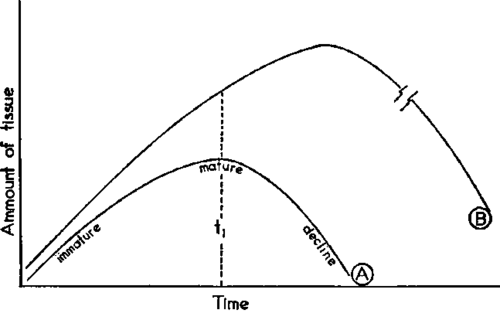
Fig. 2. Comparison of Growth Curves of Normal (A) and Neoplastic (B) Tissue
At time t1 when normal cells are in the mature state (curve A, Figure 2) only immature cells will be found in the new reaction where the cell-synthesizing system is enhanced (curve B). As the catabolic enzymes cannot degrade cells at the same rate at which immature cells are produced, the normal steady state is no longer possible, and an accumulation of young immature undifferentiated cells results. This situation is found in leukemia.
A second possible explanation, and one that may be more plausible, is that the rate of formation of cells may remain unaltered, but the rate of cell destruction may be markedly retarded. These normally acting anabolic enzymes form a new, relationship with the slowed catabolic enzymes, and the result is an accumulation of neoplastic tissue. New enzyme systems need not be postulated since only altered kinetics are being* considered. This may explain why no specific cancer enzyme has been found, and why certain enzymes, like catalase and xanthine oxidase, are less active in tumor tissues.
Continue to:


