2. The Greenstein Hypothesis
Description
This section is from the book "Chemistry Of Enzymes In Cancer", by Franz Bergel. Also available from Amazon: Chemistry Of Enzymes In Cancer.
2. The Greenstein Hypothesis
The second principal postulate is that of Greenstein.111 On the basis of numerous estimations of enzyme activity levels in normal and tumor tissues, mainly originating from his own laboratory, he came to the conclusion that there existed in malignant material a more or less uniform biochemical pattern, most striking in reference to enzymes but also with regard to other cellular constituents. While from a holo istic point of view one cannot ignore the latter, it is of special interest to the subject under discussion that, according to Greenstein, there was a closer similarity between the enzyme pattern of tumors, apparently independent of site, age, growth rate and host species, than between that of malignant and the corresponding normal tissues. In other words, there exists see"rajngly a 'tumor pattern,' with a convergence, biologically speaking, towards a primitive non differentiated cell type. Fig. 2 illustrates all this with convincing clarity, although the diagramatic values refer only to mouse tissue.
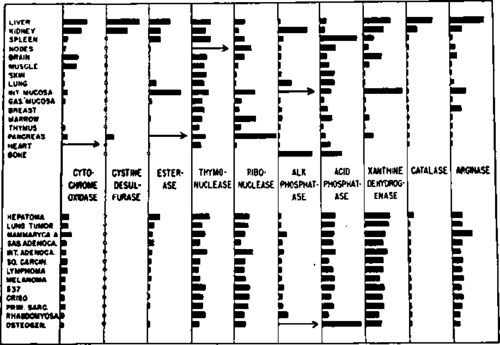
Fig. 2. Levels of various enzymes in normal and cancer tissues of mice. (From Greenstein: Cancer Res.,'" Courtesy of the American Association for Cancer Research Inc., Chicago, Illinois.).
Just as the ifs and buts of the Warburg concept were presented above, it is necessary to consider, in a similar manner, arguments for and against the Greenstein hypothesis. Its main attraction derives from its theme of uniformity which, while recognising the absence of striking qualitative biochemical differences between normal and abnormal tissues, underlines the quantitative relation between tumor and tumor. This should be helpful for everybody working on the cancer problem, because it may explain the similarity of the biological characteristics shared by nearly all neoplastic tissues, namely their unrestricted growth potentialities (although this expression according to Haddow 116117 should only be used with scientific discernment), their cytological irregularities, their invasiveness and their formation of metastases, their tendency to become de differentiated or anaplastic or autonomous, their losing cellular adhesiveness and, on an experimental basis, their gaining easy transplantability. On the other hand, as Weinhouse,259 Potter 197 and others have pointed out, there exists a diversity of biological and biochemical behaviour between malignant cells and malignant cells which are particularly apparent in the host and may be caused by complex host tumor relationships. This interplay was brought out by Greenstein 111 himself who also lectured in Japan 112 on the fact that among various chemical components in the tissue of the tumor bearing host, liver catalase was strikingly affected. Its level decreased continuously during the progressive growth of the tumor, unless the neoplastic growth was surgically removed when the enzyme regained, after a few days, its normal level. Just as in the case of catalase, the values given in Fig. 2 refer to enzyme activity levels in tissue homogenates which may or may not bear any relation to the actual amount of enzymes present in the cells, a question often raised 109 and always present in the mind of the investigator. For instance, Fraser, Kaplan and Schulman 99 have demonstrated in vitro that catalase activity could be lost by absorption of the haem enzyme on an oil water interface and could be restored by its desorption from the interface. Such and similar processes might account for changes in intracellular enzyme activity levels, and it is therefore of great importance in studies like those of Greenstein to be more certain about the meaning of enzyme assays in biological material.92 A related difficulty is brought to the fore by the effects of the analytical technique chosen for the assay of another enzyme, reported in Figure 2, namely xanthine dehydrogenase. This metallo flavoprotein 19,80 can act anaerobically as dehydrogenase and aerobically as an oxidase, except in the chicken liver, where it is only capable of acting as dehydrogenase.185,213 Greenstein's activity levels in mammalian tissues were assayed anaerobically by measuring the decoloration rates of methylene blue in tissue extracts with xanthine as substrate. His results, however, may not present a true picture of the level of activity of xanthine oxidase. That the anaerobic and aerobic activities are not always identical has been demonstrated by Richert et al.215 and Westerfeld et al.265 who claimed that certain inhibitors interfered only with the liver enzyme as oxidase but not as dehydrogenase, and did not affect the enzyme from cow's milk. Thus the situation with these enzymes of purine catabolism alone is complicated enough to warrant a solemn warning for present and future students of enzyme activities in normal and cancer tissue. Lewin,29,178 when attempting the estimation of xanthine oxidase activity levels in mammary tissue of mice, decided in favour of a method involving oxygen. Adapting Kalckar's 143 technique for determining purines, he found in contrast to Greenstein's general results in mouse organs (see Fig. 2) that the breast tissue of cancer susceptible C-strain mice (carrying the Bittner milk factor) showed about 58% relative xanthine oxidase activity (per precancerous cell) of the corresponding tissue of the cancer resistant C-strain mice (without Bittner factor) (Fig. 3). This activity falls to 25% of the latter in the mammary tumor, again calculated per cell. While these differences are impressive and significant, they are still based on activity levels assayed in vitro which as in Greenstein's series do not necessarily reflect the enzymic activity within the intact cell, since presence or absence of activators and inhibitors in vivo are as yet unknown. Although Fishman 92 stresses the limitations of cell count as a basis for enzyme estimations, the values expressed as activities per cell may be more realistic than those expressed per unit weight of tissue or homogenate, as the cellularity of the tumor (see Fig. 3) is considerably higher than those of the pre- and non cancerous tissues.
One other criticism directed towards the Greenstein hypothesis may originate with the uncertainty as to the nature of the rate limiting factors in the living organism. On the diagram (Fig. 2) it can be seen that one enzyme activity, namely that of cystine desul furase was absent in all malignant samples tested, representing what was called a qualitative difference. Since then Stekol, Weiss and Anderson 281 have demonstrated that this enzymic system consisted of two enzymes, i.e. cystine reductase and cysteine desulfhy drase (both together with xanthine oxidase to be mentioned again further on), and that the apparently low activity was not due in this case to low concentrations in the tumors of the corresponding proteinous apoenzymes but to a deficiency of their cofactors, DPNH and pyridoxal phosphate respectively. This observation rings another warning to be heeded by workers and speculators in the field of enzyme patterns of tumors: before claiming the diminution or change of holoenzymes, or of enzyme forming systems, they should ascertain whether the observed alterations in activity levels are not caused by variations in the functional amounts of coenzymes or even metal activators.
Continue to:


