Nucleic Acids. Part 2
Description
This section is from the book "Chemistry Of Enzymes In Cancer", by Franz Bergel. Also available from Amazon: Chemistry Of Enzymes In Cancer.
Nucleic Acids. Part 2
If one equates cell growth and division with the synthesis of DNA, it needs explaining why neoplastic cells appear to proceed in an "irrepressible" manner, as Potter indicated,108 with continuous DNA synthesis unless the cell dies (necrosis, etc.). It could be due to a surfeit of the anabolising enzymes of nucleic acid metabolism (see Figs. 5 and 6) or to a partial or total deficiency in some of the catabolising enzymes. A number of teams have investigated this intriguing question from different angles and have come to the provisional conclusion that mainly catabolism of nucleic acids is diminished at the level of nucleotides, nucleosides, purines and pyrimidines. This, in spite of a possible increase in some anabolic enzymes, fits in well with the 'deletion hypothesis', and if coupled with Potter's108 positive and negative feedback mechanism, might assist in solving the challenging puzzle.
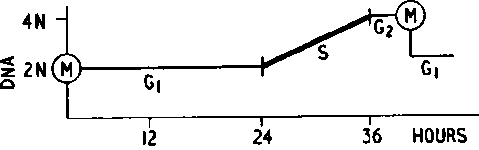
M = Mitosis
2N = Diploid amount DNA 4N = Tetraploid amount DNA S = Period of DNA synthesis G1 = First long qap } No DNA Synthesis. G2 = Second short gap }No DNA Synthesis
Fig. 9. The cell cycle in bone marrow. (From Lajtha: Physiol. Rev. Courtesy of the American Physiological Society, Washington, D.C.).
It is now necessary to give a few examples which will illustrate this thesis. With regards to purine derivatives, two approaches, one of a dynamic and the other of a more static nature, have given similar results. Bennett, Skipper et a/.,20 when using labelled metabolites in mouse sarcoma 180, in human cancer cells growing in tissue culture and Ehrlich ascites tumor, have come to the general conclusion that the pool of soluble purine derivatives was maintained at a level manyfold greater than that to be expected after taking into account the considerable extent of nucleic acid synthesis required for the increase in cell numbers (see Daoust et al.78 claiming that the rate of DNA phosphorus production was twice that of cell formation in normal lung tissue). As rapidly growing cells, such as sarcoma 180 and human tissue cancer cells, conserved apparently their DNA, RNA and soluble nucleotides, and only RNA was associated with a certain feedback to soluble precursors, the authors came to the conclusion that their detection of a nucleotide accumulation, as a possible controlling factor in the rate of cell division, could be best explained on the basis of a deficiency In catabolising enzymes. This postulate is strongly supported firstly by results of the group at the Montreal Cancer Institute, de Lamirande, Allard and Cantero156 who found that the activities of the catabolic enzymes of the 'purine cycle' in Novikoff hepatoma, when compared with those in normal liver, were either greatly diminished (5'-nucleotidase, nucleoside phosphorylase, guanase and adenase) or in the case of xanthine oxidase and uricase not demonstrable. Only adenosine deaminase activity was increased (see Fig. 10). Particularly the apparent absence of xanthine oxidase which controls the only exit from the 'purine cycle' (see Fig. 7), unless there are oxidative pathways from inosinic acid23 or xanthylic acid operating, is of great interest. It tallies well with the observation by Lewin et al.,178 as mentioned above, namely the lower level of this enzyme in the pre-cancerous and cancerous breast tissue of mice. The meaning of this was also discussed by the writer and his colleagues 28 in connection with some other work on xanthine oxidase which will be considered in more detail further on. A recent contribution by Feigelson, Ultman et al.91 disclosed a similar state of affairs in an experimental mouse leukemia L1210, where xanthine oxidase and uricase activities were diminished in comparison with normal lymphoid cells. Indirect evidence for the lack of uric acid-forming enzymes in tumors comes also from studies by Moore and Le Page184 on the metabolism of the anti-tumor drug thioguanine. They found that tumor cells were less capable of degrading the drug to thiouric acid in comparison with normal tissues. Before going any further the usual warning has to be uttered that the objects of analysis refer in the main to experimental tumors and the results may have only a restricted meaning as to the properties of neoplasms in other species. From preliminary work it appears that breast tissue of human origin, whether of normal or cancerous character, shows little xanthine oxidase activity. However, other catabolic enzymes of purines and their derivatives have to be taken into consideration and submitted to further studies. Even in the normal organs of animals there are differences in levels of xanthine oxidase activity, due to age and nutritional state.264 The livers of newborn rats show very low levels while in older, rats the enzyme levels are much higher. A similar picture was obtained when the author and his collaborators 32 assayed the content of rat livers for molybdenum, one of the metals of xanthine oxidase. They found little in fetal and newborn livers, the amounts of the metal increasing in later weeks and falling to low values in some hepatoma tissues.
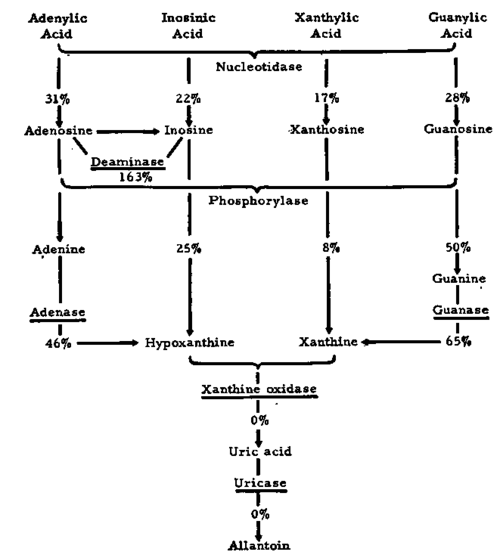
Fig. 10. Changes in purine catabolising enzyme activities of the Novikoff hepatoma, compared with normal rat liver activity as 100% (enzymes underlined) . (After de Lamirande et al.: Cancer Res.'" Courtesy of the American Association for Cancer Research Inc., Chicago, Illinois.).
From a number of publications it appears that the situation concerning pyrimidine metabolism is comparable with that obtaining with purine synthesis and degradation. The evidence for a deficiency of catabolic enzymes for pyrimidines in tumors rests on the following results:199 Pyrimidines (see Fig. 11) as such, except for orotic acid, are little if at all utilised by the mammalian organism, although Canellakis 65 has shown more recently that uracil when present in high concentrations is taken up by rat liver for polynucleotide synthesis. However, this author following the work by Rutman et al.220 has shown that mouse hepatoma in contrast to normal liver was more efficient in converting the precursor into RNA-uridylic acid. He came to the conclusion that this was probably due to the decreased capacity of the tumor to catabolise this pyrimidine or its dihydro-derivative to β-ureido propionic acid. In other words there existed a reverse relationship between the ability to degrade uracil and the capacity to incorporate it into RNA. Reichard and Skold,212 going one step further, demonstrated that in Ehrlich ascites tumors apparently no enzymes of uracil catabol-ism were present, whereas they could be detected in normal liver. Preliminary results from Potter's laboratory 200-202 indicate some differences in the metabolism of thymidine between normal and regenerating liver and Novikoff hepatoma. However, when the latter and several other primary and transplanted rat hepatomas were compared 202 with the Dunning hepatoma for deoxycytidylic deaminase (deoxycytidylic acid→ -deoxyuridylic acid-→ thymidylic acid ←-thymidine), it was found that the Dunning tumor, like normal liver, was deficient in this enzyme. Pitot and Potter198 came to the tentative conclusion that in spite of the resemblance of the Novikoff and Dunning hepatomas with respect to their deficiency in pyrimidine catabolising enzymes, their cells might derive from different normal liver cell types.
Continue to:


