Some Preliminary Results Obtained In Other Lymphatic Tumors
Description
This section is from the book "Symposium Phenomena Of The Tumor Viruses", by U.S. Dept. of Health. Also available from Amazon: Tumor Suppressing Viruses, Genes, and Drugs: Innovative Cancer Therapy Approaches.
Some Preliminary Results Obtained In Other Lymphatic Tumors
In the past, many attempts to transplant so-called spontaneous lymphatic neoplasms, which arise in older Swiss mice, were unsuccessful. However, within the last year, at least 5 tumors have been found that are readily transplantable to mice of this randombred strain; 2 of these 5 have now been examined under the electron microscope. Particles have been found in both, and the other 3 remain under study.
In view of this phenomenon, the observations in one of these tumors are presented: In a group of Swiss mice inoculated with leukemia-virus nucleic acid, a lymphoblastic lymphoma arose in 1 of the mice 5 months after treatment. This lymphoma is now in its tenth serial generation and is being followed throughout by electron microscopy. The material presented here illustrates the findings obtained in specimens from the first three generations.
The electron microscope study of this transplantable lymphoma shows an extremely large number of intracytoplasmic doughnut-shaped particles (figs. 6, 7, and 8) that do not resemble the leukemia virus. These particles, the diameter of which is about 800 A, form large inclusion bodies frequently in the Golgi zone and sometimes in contact with the nuclear membrane (fig. 7). Although the Swiss mice are generally thought to be free of the milk factor, these viruses are morphologically identical with the intracytoplasmic particles of the mouse mammary carcinoma. However, in the lymphoma cells, this type of intracytoplasmic particle is not associated with the extracellular particle consisting of four concentric membranes usually found in the mammary tumor.
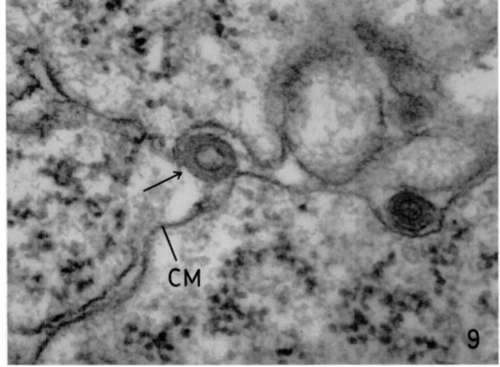
The relationship of these particles to the leukemia virus and to the etiology of this lymphoma is still not known, since it has not as yet been possible to transmit the disease to adult mice with cell-free filtrates. Finally, it remains unclear whether this transplantable tumor was spontaneous in origin or related to the experimental procedures. It must be stressed, however, that among the lymphoma cells typical mature leukemia viruses are occasionally observed in intercellular spaces (fig. 9). Obviously this is a complex problem requiring further integrated biological and morphologic studies.
It is worth noting that particles similar to those observed in the lymphoma have also been observed in a mammary tumor of a DBA mouse by Dr. Dan Moore, and in a lymphoma in a Swiss mouse by Dr. A. J. Dalton and Dr. Alice Moore.
In conclusion, with the precise morphologic techniques in electron microscopy, it is now possible to differentiate the oncogenic viruses and to look forward to their classification on the basis of morphologic data. Within such a framework it would appear that the Swiss mouse leukemia virus differs from the following viruses: Rous sarcoma, chicken leukosis, Shope fibroma and papilloma, polyoma and mouse mammary carcinoma, as well as from the lymphoma particle. It resembles, however, the virus described by Adams and Prince (11) in the Ehrlich ascites tumor, and the agent described by Graffi (12) in a mouse myeloid leukemia. It is also similar to the Gross (18) and Moloney (14) leukemia agents.
In summary: The Swiss mouse leukemia virus was studied with the electron microscope. Lead hydroxide electron staining was found most helpful for the demonstration of virus fine structure. Special consideration was given to the successive steps of virus maturation by a process of budding at the level of the cell membrane and to the different localizations of the virus. Recent observations on particles of approximately 800 A in size observed in a transplantable lymphoblastic lymphoma were also discussed.
Plate 44
Figure 1. Leukemic solid tumor. Lead hydroxide staining. At level of cell membrane (CM) of leukemic cell (L), virus bud is recognizable. Outer virus shell (( )) and inner virus shell (I) can be seen at this early stage. One mature virus (V) is seen in intercellular space. X 217,000.
Figure 2. Liver of leukemic mouse. Lead hydroxide staining. Liver highly infiltrated with leukemic cells. A further step in differentiation of virus is seen. Particle is connected to cell membrane (CM) by large pedicle (P). X 180,000.
Figure 3. Spleen of leukemic mouse. Lead hydroxide staining. A completely formed virus is seen. Maximum electron density appears between outer (O) and inner (I) shells, the central part of virus showing only a moderate electron density. This virus is still attached to cell membrane (CM) of a leukemic cell (L) by an extremely thin pedicle (arrow). X 225,000.
Figure 4. Spleen of leukemic mouse. Lead hydroxide staining. Three viruses are seen in intercellular space. In two, the section has passed through central portion of particle and therefore their centers appear clear. Another virus is seen cut more tangentially. High density of inner virus shells (I) is observable, and in 1, intermediate virus membrane is seen between two virus shells (arrow). X 220,000.

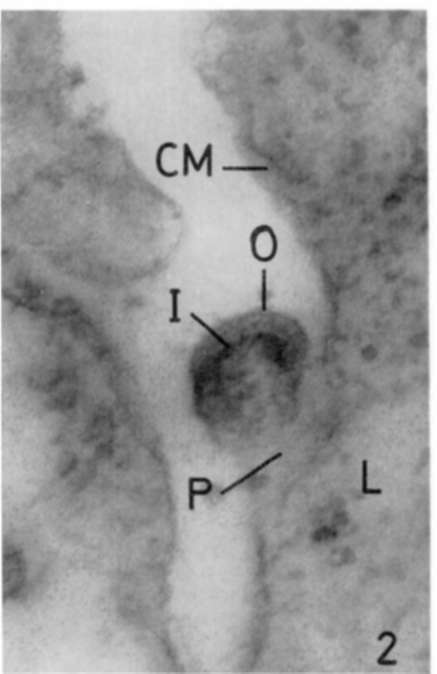
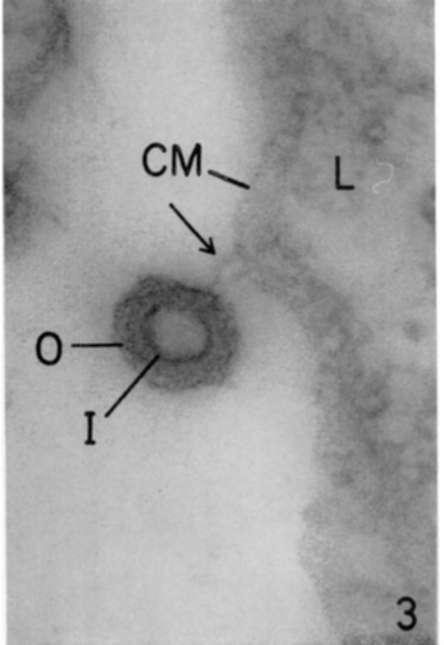
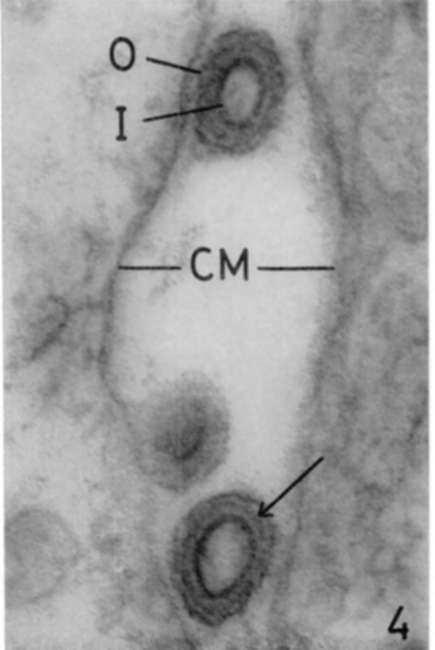
Plate 45
Figure 5. Bone marrow of leukemic mouse. No lead hydroxide staining. In cytoplasm of megakaryocyte numerous vacuoles are seen containing many viruses. These viruses appear to be budding (arrow) from membrane limiting cytoplasmic vacuoles (M) by same process observed at level of cell membranes of leukemic cells. Structure of these viruses is identical to that of viruses observed in intercellular spaces of leukemic organs. The only particularity is the frequent occurrence of dumbbell (DP) or chainlet (CP) profiles resulting from the continuity between outer shells of several viruses. X 84,000.
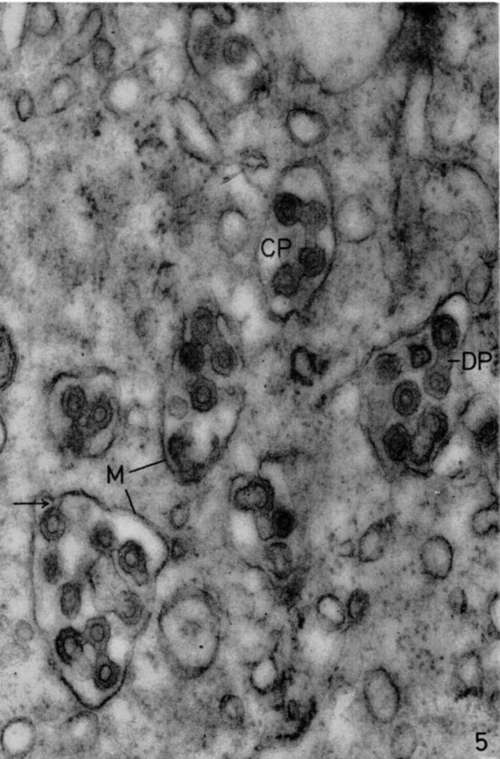
Plate 46
Figure 6. Involved thymus of mouse with lymphoblastic lymphoma. Uranyl nitrate staining. Mitotic figures arc present (MI). In at least two cells an inclusion body formed by numerous particles is seen (arrow). X 5,500.

Plate 47
Figure 7. Lymphoblastic lymphoma from subcutaneous site of implantation. Lead hydroxide staining. Near nuclear membrane (NC), group of particles extremely regular in size is seen. These particles seem to have a close relationship to nuclear membrane (arrow). X 72,000.
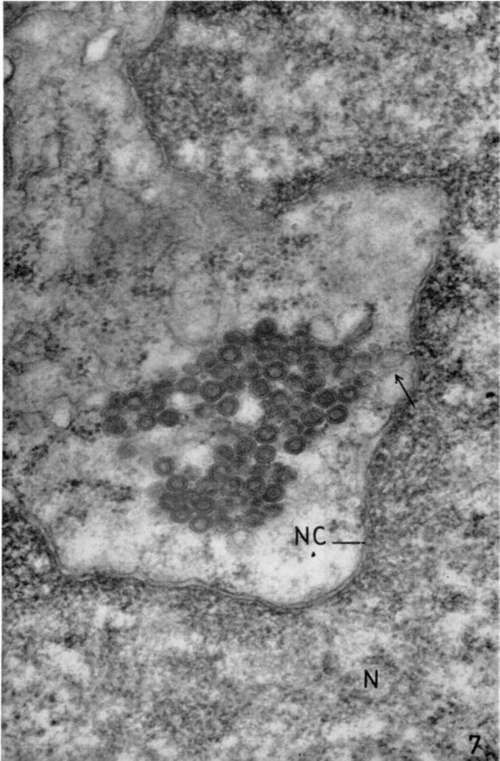
Plate 48
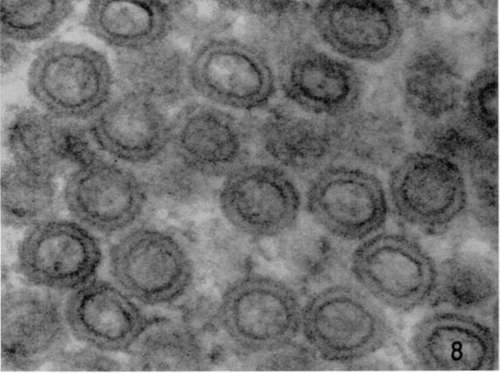
Figure 8. Lymphoblastic lymphoma from subcutaneous site of implantation. Lead hydroxide staining. High-magnification micrograph illustrating regularity of particles observed in lymphoblastic lymphoma and differences in shape between these intracytoplasmic lymphoma particles and the extracytoplasmic leukemia virus of Swiss mice. X 306,000.
Figure 9. Lymphoblastic lymphoma involving mesentery. Lead hydroxide staining. Between lymphoblastic cells of this tumor, rare but typical leukemia viruses are seen in intercellular spaces (arrow). X 145,000.
Dr. Fogh (New York State Health Department): It has been striking to me that in this otherwise wonderful seminar the information concerning work with human cells has been very limited. I would like to discuss recent results from my work with human amnion cells in tissue culture.
We found 4 years ago that cultures of amnion cells, like cultures of many other normal cells, could change to a type of cell which, in many respects, had the characteristics of malignancy. Since then we have been looking for some means of measuring quantitatively the "transforming" capacity of a culture, and we have recently had some quite encouraging results.
It was first observed that in cultures, transferred 3 weeks after the original seeding, cell colonies could develop in the transfer culture. At first the colonies consisting of changed cells were visible only when the cultures were observed in the microscope. As they grew out, they could be seen macroscopically and could be counted easily. The cultures, each originating from a seed of two million cells, contained an average of approximately 200 colonies of changed cells. There was a clear distinction between the colonies of changed cells and the degenerating primary cells which could be observed in the spaces between the colonies.
Continue to:


