Studies On Broken Cell Preparations. Part 3
Description
This section is from the book "Symposium Phenomena Of The Tumor Viruses", by U.S. Dept. of Health. Also available from Amazon: Tumor Suppressing Viruses, Genes, and Drugs: Innovative Cancer Therapy Approaches.
Studies On Broken Cell Preparations. Part 3
In summary: Further studies have been made of the ultrastructural characteristics of the myeloblasts of avian myeloblastosis. Cells were examined immediately after removal from the diseased bird, and the progressive structural changes occurring quickly in the cells in tissue culture were followed. As in earlier work, there was little evidence of difference, in electron micrographs of thin sections, between the structure of the myeloblast in the circulating blood and that of cells identified as myeloblasts in normal bone marrow. Within 2 hours of culture, under proper conditions, however, there was observed the development of many highly osmophilic spheroidal bodies, viroplasts, which have been identified as sites of infection and synthesis of the virus. During the first 4 to 6 days these structures exhibited characteristics which differed from those of cells longer in culture. After this period, there was seen, with considerable uniformity, the characteristic sequence of structures which varied from osmophilic bodies of homogeneously distributed internal substance to others which contained virus particles in small numbers and lesser amounts of material, finally, to vacuoles regarded as the organelle eliminating the agent from the cell.
Techniques For Fixation Of The Cells For Cytochemical Study By Light microscopy have been greatly refined. Additional studies have been made to identify organelles containing adenosinetriphosphatase in normal myeloblasts and their derivatives in the nondiseased bird and in the virus-associated myeloblasts from the bird and from tissue culture. Earlier observations of the enzyme in the granules of normal myelocytes and of the viroplasts of the diseased cells have been confirmed and extended. With the additional information, it has been possible to perceive possible cell-virus relationships by means of which the cell becomes involved with the virus and evidence of the contribution of normal cell processes to the multiplication of the agent. These have been indicated diagrammatically as a summary of the findings and of the hypotheses which have evolved from the studies thus far made.

Plate 38
Figure 4. Myeloblast from growing culture 2 days old. Some of mitochondria (M) exhibit normal appearance while others show evidence of swelling. There are several viroplasts (PL) some of which contain virus particles (VP). One viroplast (PLV) contains several virus particles. Structures designated (LB) are probably lipide bodies. X 30,000.
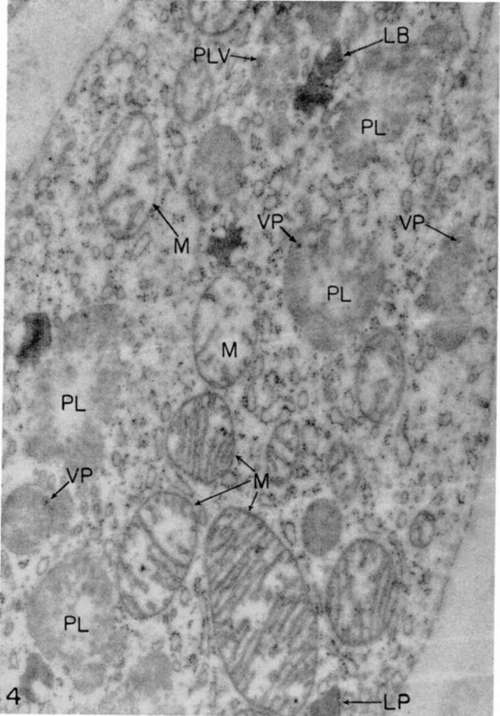
Plate 39
Figure 5. Portions of 2 myeloblasts from a growing culture 8 clays old. Several stages in postulated viroplast sequence (sec text) are seen. These include a mitochondrion of normal features (M); a mitochondrion (AM) with fewer cristae and much amorphous material; altered mitochondria (AM) bounded by double outer walls and with only suggestions of presence of cristae: viroplasts (PL) with contents of variable homogeneity, 1 containing a virus particle (VP); and, finally, clear or almost clear vacuoles (V). X 26,000.

Plate 40
Figure 6. Section through edges of 3 myeloblasts (MY1, MY2, MY3) from a growing culture 12 days old showing an unusually large number of virus particles in an intracellular bounded structure (IB) at periphery of cell (MY2). A limiting membrane (IM) can be seen about this structure and lying within cell membrane (CM). Many other virus particles (VP) lie between cells. Two mitochondria (M) are also present. X 61,000.

Plate 41
Figure 7. Mitotic figure of mycloblast from growing culture 2 days old. The spindle fibers (SF) extend from cent Holes (C) toward chromosomes (CH). Note presence in cell of several typical early-form viroplasts (PL), 1 of which contains a virus particle (VP). X 19,000.
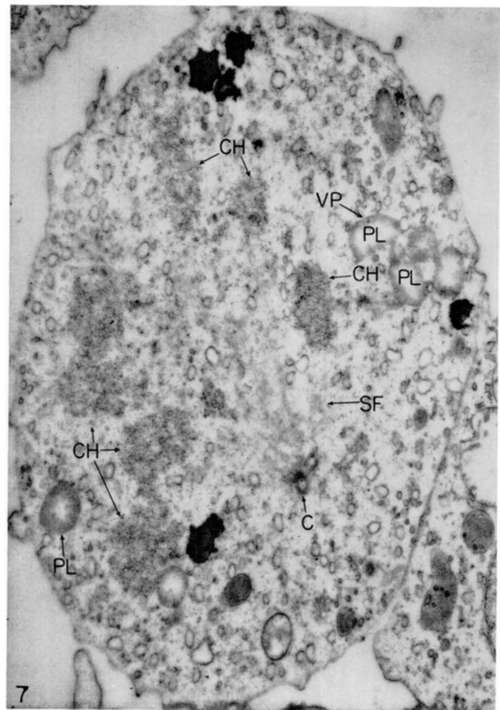
Plate 42
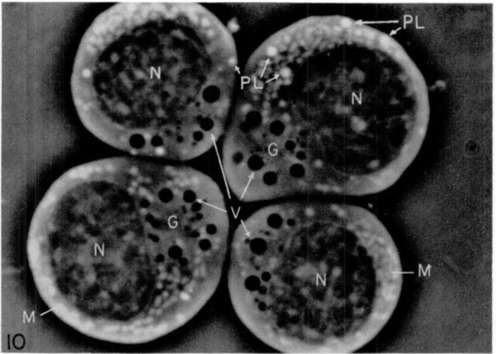
Figure 8. Phase-contrast photograph of living cells from bone marrow of normal (uninfected) chicken. The respective ceils were identified as a heterophil (HP); 2 myelocytes (MCL); and 3 mycloblasts (MB). Black structures (G) represent granules and white forms (V) are vacuoles. X 3,200.
Figure 9. Same cells as those of figure 8 after freezing, dehydration, fixation, and treatment with Wachstein-Meisel reagent for staining of adenosinetriphosphatase (see text). Symbols are same as those of figure 8. There is dense staining of some, but not all, of the granules of the heterophil and of the myelocytes. X 3,200.
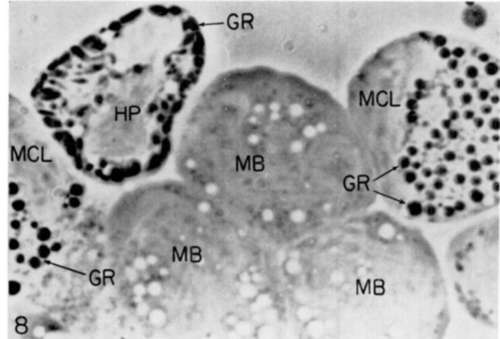
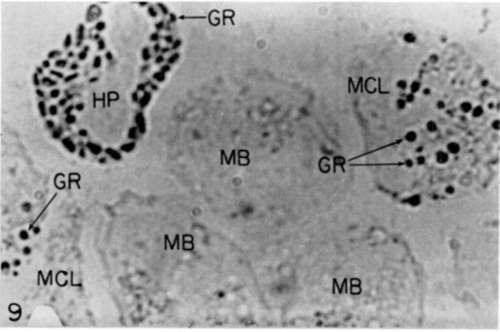
Plate 43
Figure 10. Phase-contrast photograph of living cells from growing culture of myeloblasts 61 days after transfer from circulating blood of chicken with myeloblastosis. Nuclei (X) are large and show indications of a nuclear notch. In these regions is the Golgi zone (G) surrounded by vacuoles (V). Mitochondria (M) are distributed through the cytoplasm with few in Golgi region. Several bright structures are identified as viroplasts (PL). X 3,200.
Figure 11. Same cells as those in figure 10 after freezing, dehydration, fixation, and treatment with Wachstein-Meisel reagent for staining of adenosinetriphosphatase (see text). Symbols are the same. Many of viroplasts (PL) (bright bodies of figure 10) are heavily stained with lead. X 3,200.

Discussion
Dr. Beard: Before beginning the discussion of Dr. Bonar's presentation, concerned with the discovery of the ATPase activity of the granules of normal myelocytes and granulocytes and of the general technique for the studies by light microscopy, it should be mentioned that the various sequences were devised by Dr. Mike Haddad (St. Francis Hospital, Hartford) as described in his paper now in press.
Dr. Gey (Johns Hopkins Hospital): Would it not be possible to consider that the material in the viroplasts and vacuoles was being taken up rather than being eliminated by the cells?
Dr. Honar: From the morphologic evidence, it could not be said definitely that the material was being eliminated from the cell, because the single photographs are fixed in time. However, when the morphologic evidence is correlated with that from tissue culture, it seems that there is no other explanation. Otherwise, masses of virus should be seen inside the cell.
Dr. Gey: If that is true, then it should be possible by phase microscopic examination to sec them going out. Has this been observed?
Dr. Bonar: This aspect of the problem is now under study. Of course, only the viroplasts and vacuoles could be seen in the phase microscope, not the virus particles themselves.
Dr. Francis (University of Michigan): Would you explain the function of the viroplast in the production of virus in the myeloblasts that is unlike the functions of the vacuoles Dr. Haguenau observed, in which virus was seen in Rous sarcoma cells? It seemed to be her view that the vacuoles and the particles in them were the result of pinocytosis.
Dr. Bonar: Again there arises the problem of interpreting a dynamic process from static pictures. It is possible that some of these vacuolar inclusions in the myeloblasts do arise by phagocytosis or pinocytosis of virus particles, though, personally, I think this is probably not the case.
We believe that the virus particles seen in the vacuoles in the myeloblasts are derived from the viroplasts, because there is evident the range of structures apparently involved. This begins with bodies with a dense, rather homogeneous matrix, in which one can occasionally see faint outlines of virus. From these dense bodies there is a spectrum of structures including those with distinct virus particles in a dense matrix, those which have lost part of their nonviral content, and, finally, the vacuoles which contain virus particles and little else.
While it is possible that some virus particles might have reached vacuoles by the process of phagocytosis, it seems extremely unlikely that the process could be reversed all the way to the structures with a dense matrix with only one or a few virus particles embedded in this matrix.
With respect to Dr. Hagucnau's pictures, there is the same difficulty of interpretation. I suspect that our gray bodies arc similar to the rather dense structures containing virus particles which she has seen in Rous sarcoma cells, and that they are probably analogous to the altered mitochondria containing virus particles which Benedetti and Bernhard have observed in erythroblastosis. We have not had the good fortune to sec virus particles in a really recognizable mitochondrion in myeloblasts, though we have searched diligently. It may be, of course, that virus particles do not occur in recognizable mitochondria in myeloblastosis.
Dr. Haguenau: From Dr. Bonar's pictures I would be convinced that he is right in the assumption that these particles, though we cannot say anything morphologically, arc being extruded from the myeloblasts, because he has other evidence of the process. I was impressed that he did not have (at least in the pictures he has shown and, of course, he did not choose them for that) any of this amazing pinocytosis activity that we have seen together with extraordinary pseudopod activity of Rous sarcoma cells, which I have not illustrated directly. Nevertheless, this is very striking. I am not sure that the situation with Rous sarcoma system is absolutely comparable with that of myeloblastosis.
Continue to:


