Studies On Broken Cell Preparations
Description
This section is from the book "Symposium Phenomena Of The Tumor Viruses", by U.S. Dept. of Health. Also available from Amazon: Tumor Suppressing Viruses, Genes, and Drugs: Innovative Cancer Therapy Approaches.
Studies On Broken Cell Preparations
In earlier studies, it was found (34) that when myeloblasts from non-growing cultures were washed with saline and disrupted by freezing and thawing, a high adenosinetriphosphatase activity was found in the homogenate. When cell-free homogenates exhibiting high enzymatic activity were injected into chickens, myeloblastosis was not induced, though the amount of enzyme activity was 10 times more than that necessary to have caused infection if it had been associated with the typical virus particle. At that time there was no information as to the possible state of the enzyme. The results of electron and light microscopy obtained later, however, suggested that most, if not all, was present in the walled structures, gray bodies, or viroplasts. To test this possibility, myeloblasts from growing cultures were disrupted and the homogenate was fractionated by centrifugation at different speeds. By this means it was observed that the specific enzymatic activity per gram of protein was highest in the fraction coming down at speeds used for the sedimentation of mitochondria. The value observed was related to the characteristics of the cells studied, but it was always much higher than the adenosinetriphosphatase activity of normal mitochondria (42, 43).
Discussion
The results of the present investigations, together with those of related studies, are of principal significance in their bearing on the clarification of certain limited aspects of host cell response to the virus of avian myeloblastosis. For purposes of discussion, cell response, in the sense intended, may be regarded as the development (1) of structures physically and chemically identifiable as specific to the intracellular synthesis and subsequent elimination of the virus from the cell and (2) of processes inimical to the progressive maturation of the myeloblast in the normal pattern but compatible with continued and unrestrained growth, which is the major attribute of malignancy. Interpretation of the findings may be aided somewhat by consideration of some characteristics of the normal myeloblast and the extrapolation of the concept of the nature and potentialities of the uninfected cell to the explanation of the behavior of the myeloblast after invasion by the virus. Actually, the cell of primary susceptibility to the agent has not been identified. Inoculation of virus into the intact chicken results in the occurrence of the agent in reticular cells of the spleen and bone marrow, macrophages, and endothelial cells in the liver as discerned in thin sections (44, 45), and, later in the disease, of other cells or tissues (46). Myeloblastic leukemia, per se, however, is manifested as a malignant disease only by the overgrowth of myeloblasts. This could result by direct infection of the myeloblast in its most primitive stage before the onset of the maturative processes, but the possibility must also be considered that primary infection may take place in the precursor of the myeloblast and that the virus is passed to the derivative daughter myeloblasts. The studies on infection of normal bone marrow in vitro (24) provide no unequivocal clarification of this question, since the tissue studied probably represented not only myeloblasts but other bone marrow cells as well. It must be recognized, nevertheless, that once myeloblasts are invaded by the virus, they become the representative proliferating cell, and, in consequence, the following discussion is based on the assumption that the myeloblast is the cell of primary response to the agent. The principal points for discussion are illustrated in the diagram of text-figure 1, which is concerned with those organelles thus far known or thought to be involved in myeloblast response to the myeloblastosis virus.
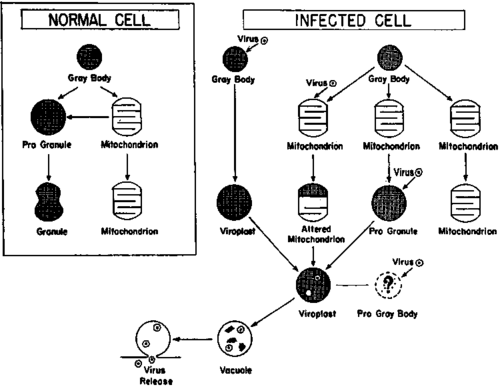
Text-figure 1. Diagrammatic representation of potential derivatives of gray bodies (microbodies) in the normal myeloblast and in myeloblasts in association with the virus of avian myeloblastosis.
The myeloblast, as the term implies, is not an end-form cell but simply a primitive element transitional between the precursor hematopoietic tissue and the mature polymorphonuclear leukocytes of the circulating blood. As such, the cell may multiply under normal conditions, as shown by mitotic figures in stained preparations of bone marrow, or begin maturation into the myelocytic series marked by the appearance and progressive increase in the number of granules. These structures, which appear to be of major significance for the present work, are of uncertain origin, but some evidence indicates that they may arise from mitochondria (40, 41)- In turn mitochondria have been thought to be derived (82, 88, 89), at least in part, from microbodies, or as designated here, gray bodies. It may be, also, that mitochondria may arise by segmentation of preexisting mitochondria. At the left of text-figure 1, these possible sequences have been indicated diagrammatically.
There is no reason to suppose that the principles of potential organelle derivation differ in the infected cell from those of the normal myeloblast. A major difficulty in the interpretation of the nature of cell response resides in the uncertainty of distinguishing the primary site of virus activity. At the right of text-figure 1 are indicated 4 possibilities, for each of which there exists an experimental basis.
1. The best evidence of the site of virus involvement is that concerned with the gray body. This is the structure present in the myeloblasts in the circulating blood of the diseased chicken and the structure that exhibits increase in number and size during the first hours when the cells are transferred to tissue culture. The structures transitional from the gray body to the various forms of viroplasts and, finally, to vacuoles containing virus particles, as indicated in the first sequence in the diagram to the right, can be followed clearly in thin sections of cultured cells. In this cycle it would appear that the gray body is one primary site of infection. There is no information as to the nature of the virus substance which invades the cell, that is, whether it is the whole virus or bare nucleic acid, as known for other agents (47). In the diagram the use of symbols resembling whole virus particles is not meant to indicate a specific concept.
2. In the second sequence it is suggested that the mitochondrion, during the process of or immediately after derivation from the gray body (micro-body), may be invaded by the virus particle or the infectious moiety of the agent. For this there is considerable evidence in the frequent occurrence of structures with the appearance of residual cristae apparently in the course of alteration under the influence of the virus.
3. A third potentiality is that infection may occur in the stage of transition of the mitochondrion to the granule or progranule, if we assume the validity of the hypothesis that granules may be derived from mitochondria (40, 41). There is no direct evidence for this, but it is reasonable to consider a connection on the basis of the cytochemical findings, which show that both viroplasts and normal granules are rich in adenosine-triphosphatase.
4. Finally, there is some evidence to suggest that the infecting virus component may become integrated with undefined cell substance (pro-gray body) which subsequently becomes segregated and walled and exhibits in thin sections the characteristics of the gray body or viroplast.
Continue to:


