Leucin ((Ch3 )2: Ch.CH2.CHNH2COOH)
Description
This section is from the book "The Elements Of The Science Of Nutrition", by Graham Lusk. Also available from Amazon: The Elements of the Science of Nutrition.
Leucin ((Ch3 )2: Ch.CH2.CHNH2COOH)
Present in all proteins. Convertible into β-oxybutyric acid.
Leucin when given to a phlorhizinized dog produces little or no glucose.2 When added to a perfusing fluid and passed through a surviving liver leucin yields acetone bodies in large amounts.3 Baer and Blum4 found a greatly increased output of β-oxybutyric acid after giving 33.7 grams of leucin to a diabetic patient. The chemical reaction undoubtedly follows the known laws of oxidation on the α-amino group of the amino-acids of β-oxidation and cleavage of a methyl radicle whose further fate is unknown. These reactions may thus be presented :

The end-product of the metabolism of leucin is, therefore, the same as the end-product of ordinary fat metabolism.
Phenylalanin, C6H5. CH2. CHNH2 COOH, and Tyrosin, HO.C6H4.CH2.CHNH2COOH - Yield β-oxybutyric acid, and in alcaptonuria homogentisic acid. Present in all proteins, except that tyrosin is absent in gelatin.
The metabolism of these substances has already been considered in some detail (see p. 178). Embden and Baldes5 state that when phenylalanin is added to the perfusing fluid passing through the liver it may be converted into tyrosin. Even though phenylalanin does not always yield tyrosin in the organism, yet it is believed that it may be converted into p-oxyphenylpyruvic acid, which is the first oxidation product of tyrosin.
1 Dakin: "Journal of Biological Chemistry," 1913, xiv, 321.
2 Halsey: "American Journal of Physiology," 1904, x, 229; Dakin: Loc. cit.
3 Embden, Salomon, and Schmidt: Loc. cit.
4 Baer and Blum: "Arch. f. ex. Path, und Pharm.," 1906, lv, 89.
5 Embden and Baldes: "Biochemische Zeitschrift," 1913, lv, 301.
In the phenomenon called alcaptonuria (see p. 178), tyrosin and phenylalanin are believed to be oxidized only as far as homogentisic acid, in which form they appear in the urine. Falta1 reports that if phenylalanin or tyrosin be administered in alcaptonuria each is completely converted into homogentisic acid and so eliminated. In alcaptonuria the ratio between homogentisic acid and nitrogen elimination in the urine is quite constant, being 45 :100 or 50: 100,2 and the distribution of the various other nitrogenous compounds in the urine remains normal.
Neubauer and Falta3 emphasized the idea that homogentisic acid is always formed in normal metabolism, but in this rare disease cannot be oxidized. The power to split the benzol ring was absent.
However, Dakin4 has administered to alcaptonurics para-methylphenylalanin, CH3. C6H4. CH2 CHNH2COOH, and para-methoxyphenylalanin, CH3O.C6H4. CH2CHNH2COOH, substances which cannot undergo the quinoid transformation, (see p. 178) and has found that these are oxidized in the organism. He, therefore, concludes that the formation of homogentisic acid in metabolism is always pathologic, and that the benzol ring can be broken even in alcaptonuria without its intermediation. Fromherz and Hermanns6 believe that the aromatic amino-acids normally follow a dual path to destruction and that one of these is closed in alcaptonuria, whereas the other remains open. They present this picture of the process:
1 Falta: "Biochemisches Centralblatt," 1904-05, iii, 175.
2 Langstein and Meyer: "Deutsches Archiv fur klinische Medizin," 1903, lxxviii, 161; Schumm: "Miinchener med. Wochenschrift," 1904, li, 1599; Garrod and Hele: "Journal of Physiology," 1905, xxxiii, 205; Ravold and Warren: "Journal of Biological Chemistry," 1909-10, vii, 465.
3 Neubauer and Falta: "Zeitschrift fur physiologische Chemie," 1904, xlii, 81.
4 Dakin: "Journal of Biological Chemistry," 1911, ix, 151.
5 Fromherz and Hermanns: "Zeitschrift fur physiologische Chemie," 1914, xci, 194.

It will be recalled that muconic acid has been considered as representing the opening door of the benzol ring, ever since Jaffe1 gave benzol to a rabbit and found muconic acid in the urine.
Phenylalanin, tyrosin, homogentisic acid,2 and muconic acid3 all yield acetone bodies when perfused through a surviving liver.
The reaction involving the production of β-oxybutyric acid cannot yet be written, though two of its four carbon atoms are probably derived from the phenyl ring and two from the side chain.4
Tyrosin yields no sugar in the phlorhizinized dog,5 nor does phenylalanin.6
When the ammonium salts of the keto-acids corresponding to phenylalanin and tyrosin are perfused .through a surviving liver there may be a synthetic production of the two last-named substances.1 The reaction of deamination is, therefore, reversible in these cases.
1 Jaffe: "Zeitschrift fur physiologische Chemie," 1909, lxii, 58. 2 Embden, Salomon, and Schmidt: Loc. cit.
3 Hensel and Riesser: "Zeitschrift fur physiologische Chemie," 1913, Ixxxviii, 38.
4 Wakeman and Dakin: "Journal of Biological Chemistry," 1911, ix, 139. 5 Ringer and Lusk: Loc. cit.; confirmed by Dakin.
6 Dakin: "Journal of Biological Chemistry," 1913, xiv, 321.
1-Serin, CH2OH.CHNH2 COOH - Detected in small quantities in many proteins. Three carbon atoms are able to enter into the formation of glucose.
Dakin2 showed that the ingestion of 11.9 grams of serin by a phlorhizinized dog resulted in the excretion of 11 grams of extra glucose in the urine. One might picture the conversion of serin into glyceric acid which Ringer and Lusk (p. 193) showed is transformed into glucose:

But in order to remove the asymmetry of the central carbon atom it seems more probable that a keto-body is an intermediary oxidative product. The transformation might take the following form:
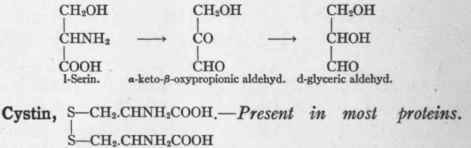
Six carbon atoms probably may enter into the formation of glucose. In a rare disease called cystinuria cystin appears dissolved in the urine, or it may take the form of stone or of sediment. If cystin be administered to a normal person it is oxidized, and does not alter the normal relation between oxidized and unoxidized sulphur in the urine.3 If cystin be given to a patient with cystinuria a part is eliminated, but a still greater part is oxidized.1 The disturbance, therefore, is not complete. When protein is given in increased measure the cystin elimination is increased in the cystinuric patient.2 The increase in neutral sulphur found in the urine is at the expense of the alkaline sulphate usually found there.
1 Embden and Schmitz: "Biochemische Zeitschrift," 1910, xxix, 423.
2 Dakin: hoc. cit.
3 Blum: "Hofmeister's Beitrage," 1904, v, 1.
In normal metabolism cystin is probably first broken up into two molecules of cystein, for on giving brombenzol mercapturic acid appears in the urine. This acid is a compound of cystein, brombenzol, and acetic acid. The reaction, as shown by Friedmann,3 takes place as follows:
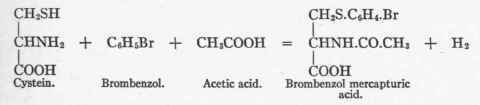
This affords an example of acetylation not uncommon in the organism.4 Acetic acid is probably constantly available, being derived from the metabolism of fat (see p. 302).
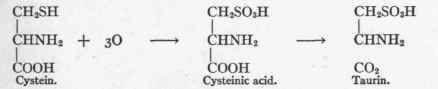
Marriott and Wolf5 further investigated this condition of artificially induced cystinuria, and were able to increase the unoxidized sulphur (cystein-S) in the urine fourfold by this means, and nearly to remove all the oxidized sulphur. The sulphur was, therefore, not oxidized to sulphate as in the normal state. That cystein is the mother substance of the taurin of the bile Friedmann6 illustrates in accordance with the following formula:
1 Williams and Wolf: "Journal of Biological Chemistry," 1909, vi, 337.
2 Alsberg and Folin: "American Journal of Physiology," 1905, xiv, 54. 3 Friedmann: "Hofmeister's Beitrage," 1904, iv, 486.
4 Consult von Furth: " Probleme der physiologischen und pathologischen Chemie," Leipzig, 1913, ii, p. 465.
5 Marriott and Wolf: "American Medicine," 1905, ix, 1026. See also Zeller and Straczewski: "Archiv fur Physiologie," 1914, p. 585.
6 Friedmann: "Hofmeister's Beitrage," 1903, iii, 1.
Taurin, however, is not the only pathway of cystein destruction, for Dakin1 gave a phlorhizinized dog 15.7 grams of cystein, which theoretically is convertible into at most 11.6 grams of glucose, and found 12.2 grams of extra glucose in the urine. This transformation would be conceivable through hydrolysis and the conversion of cystein into serin, which, as already stated, is convertible into glucose.

Dakin made note of the fact that the sulphur excretion under the conditions of his experiments was in the form of sulphate and was relatively very slow. This is contrary to the usual behavior of the sulphur contained in cystein (see p. 168).
It is evident that cystein metabolism has the choice of at least two different pathways in the organism.
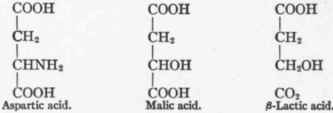
Aspartic Acid, HOOC.CH2.CHNH2.COOH - Present in most proteins. Three carbon atoms enter into the formation of glucose.
Aspartic acid was given to phlorhizinized dogs by Ringer and Lusk2 and the equivalent of three carbon atoms was recovered as glucose in the urine. This has been confirmed in Cremer's laboratory by Hering,3 who administered aspar-agin. The possible pathways of this transformation are several. Ringer and Lusk gave the following course:
1 Dakin: Loc. cit.
2 Ringer and Lusk: Loc. cit.
3 Hering: "Cremer's Beitrage zur Physiologie," 1914, i, 1.
Later Ringer1 found that malic acid was in large measure convertible into glucose.
This conception of intermediary reaction is supported by the finding of Ackermann2 that digestion of aspartic acid with putrid pancreas produces β-alanin, H2NCH2.CH2.COOH.
Dakin3 considers that alanin or lactic acid are the primary cleavage products of aspartic acid metabolism, and this is attested by the researches of Meyer4 in Neuberg's laboratory, who finds that tissue pulp of liver and muscle convert keto-succinic acid into pyruvic acid. The reaction would then take place as follows:
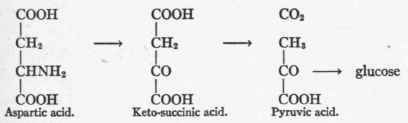
One, or perhaps both, of these pathways are open in the organism for the metabolism of aspartic acid and for synthesis of glucose from it should conditions favor.
Continue to:


