Manufacture Of Chloral
Description
This section is from the book "Alcohol, Its Production, Properties, Chemistry, And Industrial Applications", by Charles Simmonds. Also available from Amazon: Alcohol: Its Production, Properties, Chemistry, And Industrial Applications.
Manufacture Of Chloral
Chloral is trichloroacetalde-hyde, CCl3CHO, and is prepared by saturating ethyl alcohol with dry chlorine. The alcohol should be as nearly anhydrous as can conveniently be obtained - preferably commercial " absolute " alcohol. The final product of the chlorination is a crystalline mixture consisting mainly of chloral alcoholate and chloral, with some of the intermediate compounds and by-products. From this mixture chloral is obtained by distillation with strong sulphuric acid.
The reaction involved in the chlorination is a complex one. It may be explained by assuming that the alcohol is first converted into aldehyde, CH3CHO, which is then transformed into acetal, CH3CH(OC2H5)2, and subsequently into the corresponding tri-chloroacetal, CC13CH(OC2H5)2. This, by the action of the hydrochloric acid evolved, is then converted into the chloral alcoholate, CCl3CH(OH)(OC2H5).
According to another view, dichloro- and trichloro-ethyl ether are intermediate stages in the reaction, the latter compound then passing into chloral alcoholate: water to absorb the gaseous hydrochloric acid evolved. The chlorine, prepared by one of the usual industrial processes and carefully dried, is introduced very gradually, to avoid possible ignition of the alcohol. Apart from this risk, unless the initial temperature is carefully controlled, the yield of chloral is diminished through the occurrence of secondary reactions. In the early stages, therefore, the vessel is cooled; but later, when the action moderates, the carboy is gently heated, at first to 60° and finally to 100°. On the completion of the reaction the liquid becomes wholly soluble in water, and acquires a specific gravity of about 1-4. In some factories the chlorination period extends over 10 to 14 days. Certain substances which act as chlorine-carriers regularise the reaction quite notably: thus iodine in the proportion of 1 per cent. of the alcohol has been recommended for this purpose (Springmuhl).
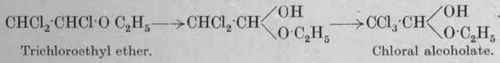
Formerly the chlorination was a tedious operation. Chlorine was passed into a relatively small quantity (25 kilos.) of absolute alcohol contained in a glass balloon, first kept cold, and then gently warmed, the process extending over weeks.2
According to A. Trillat3 the alcohol is placed in a large glass balloon or carboy, connected with one or more vessels containing
1 Ber., 1884, 17, 649.
2 Roscoe and Schorlemmer, "Treatise on Chemistry," 3, (i), 530i 3 Bull. Soc. chim., 1897, 17, 230.
The crude product is transferred to an enamelled distilling vessel (or to a lead-lined copper still) fitted with a reflux arrangement and provided with a good thermometer for fractionating purposes. An equal weight of strong sulphuric acid is added, little by little; and the mixture is then heated to the boiling-point in order to expel hydrochloric acid and other impurities, such as ethyl chloride. After the evolution of the gas has ceased, the residue is distilled until the temperature of the vapours rises above 100°. The chloral passes over at about 97°.
To remove the last traces of acid, the distillate is rectified (preferably over calcium carbonate); the fraction passing over at 94° and upwards constitutes the chloral. This is then converted into chloral hydrate by mixing it gradually with the theoretical proportion of water, namely, 12.2 per cent. of its weight. The mixture becomes warm through the reaction. It is well stirred up. and the resulting hydrate either spread out on a smooth surface and allowed to solidify, or else further purified by crystallisation from chloroform, benzine, or other solvent (see below). For example, the still warm hydrate may be mixed with one-third of its volume of chloroform, and allowed to cool in closed receptacles. After about a week the crystallisation is complete. The crystals are drained off and dried at the ordinary temperature, whilst the mother-liquor serves for crystallising further quantities.
As already indicated, the chlorination on a small scale by the older methods is a lengthy process. In recent years, however, the operations have been much shortened and cheapened by working on a larger scale and with improved methods.1
The chlorination vessels are of 2,000 to 5,000 litres capacity
1 Ullmann, " Enzyklop. der Techn. Chem." 3, 404.
(440 to 1,100 gallons), made of lead-lined iron, and fitted with coils by means of which they may be heated or cooled. They are filled two-thirds full with alcohol. The chlorine employed is purified liquid chlorine supplied in steel cylinders, or, in some cases, chlorine produced in electrolytic alkali works is used direct. It is led into the bottom of the vessel, and distributed in fine jets by means of a spreading arrangement. The supply is delivered under a gentle pressure, and any chlorine not absorbed can pass into a second chlorination vessel. In fact it is usual to instal a series of vessels, with a reflux condensing arrangement between each two, so that no chlorine is lost, and the hydrochloric acid vapours, being freed from uncombined chlorine, can be condensed and recovered as pure acid. The supply of chlorine is so regulated that the chlorination in the first vessel is finished in from two to three days.
In the early stages the temperature is kept low by means of the water coils. It is then allowed to rise, first to 50° on the second day, and finally, on the third day, to 95°. The product acquires a specific gravity of about 1.51 by the time the operation is complete, and yields at least 75 per cent. of crude chloral when a sample is tested by distillation with sulphuric acid.
The separation and purification of the chloral are carried out essentially as already described.
Chloral is an oily liquid, of characteristic penetrating odour, boiling at 97°, and of sp. gr. 1512 at 20°. A certain quantity is used for the preparation of hypnotics, but mostly it is employed in making chloral hydrate.
Continue to:


