4. Model System For Cysteine Desulfhydrase
Description
This section is from the book "Chemistry Of Enzymes In Cancer", by Franz Bergel. Also available from Amazon: Chemistry Of Enzymes In Cancer.
4. Model System For Cysteine Desulfhydrase
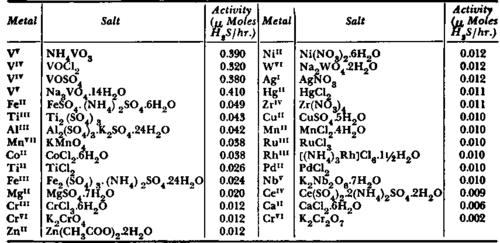
This example represents an illustration for the possible use of a model enzyme in chemotherapy and at the same time for the postulate described in previous pages, namely of increasing catabolic processes of substrates more avidly taken up by abnormal than by normal cells. When Weisberger and Levine 262 found that leukemic leucocytes of human origin appeared to incorporate cystine at a greater initial rate than corresponding healthy leucocytes, the seleno-ana-logue of cystine was prepared and studied by the same authors.268 (See Fig. 25). The writer and his colleagues (Bergel, Bray and Har-rap30) thought first of purifying cysteine desulfhydrase from rat liver, as this enzyme studied by Formageot et al.,102 Chatagner,70 Smythe 228 and others,128 catalyses the degradation of cysteine and liberates hydrogen sulfide. Unfortunately, attempts at obtaining purified preparations with high activity failed and, consequently, studies127 were directed towards the coenzyme of cysteine desulfhydrase, pyridoxal phosphate. It was hoped that in presence of metals this might act as a model for the holoenzyme, because Snell et al.220 have demonstrated that pyridoxal with metal ions, such as Al+++, Fe++, Fe+++, Cu++, etc. promoted reactions of amino-acids in vitro, namely transamination, α,β-elimination, decarboxylation and others, normally occurring only in presence of specific apo-enzymes. Subsequently, Snell 229 proposed mechanistic schemes for these reactions, which in the case of the transamination process involved the formation of a Schiff's base, chelated with the metal ion to undergo electronic displacements and to form, via a transitional intermediate, a condensation product of an a-keto-acid and pyri-doxamine. Metzler and Snell180 observed also a degradation of serine and cysteine with pyridoxal-aluminum at 100° C. and pH 5. Following this, Bergel et a/.80 found that cysteine under nitrogen with pyridoxal phosphate and an aluminum salt lost hydrogen sulfide already at room temperature. When a wider examination of transition metals was undertaken, it was revealed that vanadium salts (NH4VO3, VOCl2 or VO.SO4) with pyridoxal phosphate at 37° C. and pH 5.8 were eight to ten times more effective than ferrous salts in promoting cysteine degradation (see Table II).
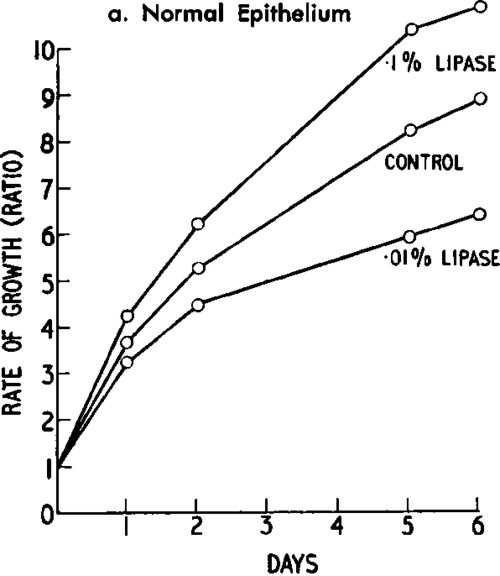
Fig. 24 a and b. Effect of wheatgerm 'lipase' on normal and cancerous tissue cultures of hamster kidney cells. (From Ambrose et al.: Exper. Cell Res' Courtesy of the Academic Press., Inc., New York.).
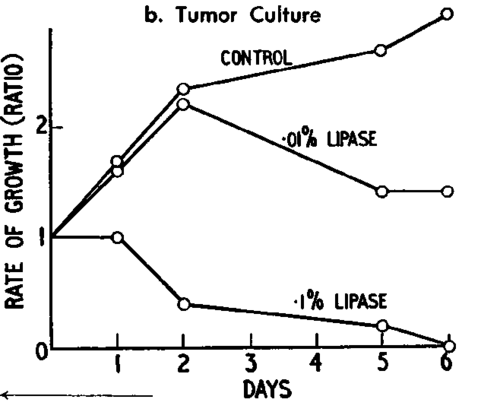
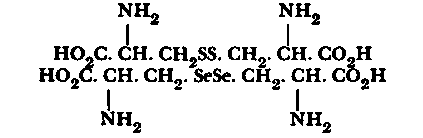
Fig. 25. Cystine and seleno-cystine formulae.
No other metal ions approached this order of effectiveness. As in the experiments of Metzler and Snell,180 it was found that among the end-products of this model catabolism of cysteine were pyruvic acid and ammonia, but amounting only to 15% of the theory, while up to 70% of the expected quantity of sulfur was liberated in form of H2S. When looking for other degradation products, it was discovered that in addition alanine, serine, cystine, and lanthionine had been formed, probably originating with the intermediate amino-acrylic acid. Further experiments revealed optimal conditions in vitro as to the ratio between cysteine, pyridoxal phosphate and vanadium salt and as to the ratio between cysteine and cystine, a maximal rate of HaS evolution being obtained when this ratio was 1:1.
Table 2
Desulfhydrase activities of various metals (0.38 mM) in combination with pyridoxal phosphate and cysteine (pyridoxal phosphate = 0.38 mM, cysteine =12 mM, temp. = 37°C, 0.1 M acetate, pH 5.8). (From Harrap et al.: Lecture Int. Congr. Bioch., Vienna. 1958."7)
It is perhaps due to these rather stringent optimal conditions that the effectiveness of this model system in vivo has so far proved to be less convincing. Elson86 observed that pyridoxal phosphate and vanadium salt ('pyrvanal' for short) exhibited a greater toxicity when injected i.p. into rats on a protein-low diet (low in cysteine-cystine) than in animals on a normal diet. In chemothera-peutic experiments pyrvanal injections did not inhibit the Walker rat carcinoma 256,118 but in mice with transplanted leukemia, originally produced by X-radiation, it increased slightly but repeatedly the survival time of the animals75 when injected daily for 10 days. No effect on survival time was noted with the lymphatic leukemia LI210 in mice,174 even when DPNH, the coenzyme of cystine reductase,231 was applied simultaneously. All these experimental animals were given an unrestricted access to mixed food. In this connection it should be remembered that Greenstein 110 reported on a lower incidence of induced leukemia in DBA mice due to a cystine deficient diet. When Harris (see Bergel et al.30) applied pyrvanal to the D-R"D" and Rous fowl sarcomas, implanted in the chorioallantoic membrane of chicken eggs, inhibition of tumor growth was observed in both cases with no effect on the embryos in the latter and with only a slight one in the former case (see Table3). It is clear that further investigations should be carried out on a number of experimental leukemias before this combination of coenzyme and metal is tried on desperate cases of acute leukemia in human patients. This is particularly desirable if naturally occurring vanadium, as trace metal, were involved together with pyridoxal phosphate and proteins in sulfur-metabolism of biological systems. This working hypothesis is supported by various reports in the literature, such as that by Snyder and Cornatzer230 which indicated a lowering of the sulfhydryl level in the liver of rats, following intraperitoneal injections of vanadyl sulfate.
Table 3
Effect of pyrvanal on weights of chick embryos, and fowl sarcomas in the chorioallantoic membrane of the chicken egg (average weights expressed as percentage of control data. Dose: pyridoxal phosphate 1.11 mg. and ammonium vanadate 0.04 mg. in 0.25 ml. water; controls on 025 ml. of saline). (After Harris et al.: see Bergel et al.: Nature." Courtesy of Macmillan and Co. Ltd., London.)_
DR"D" | Rous | ||||||
Control | Pyrvanal | Pyridoxal Phosphate Alone | NH4VO2 Alone | Control | Pyrvanal | NH4VO2 Alone | |
Embryos | 100 | 79 | 82 | 100 | 100 | 100 | 96 |
Tumours | 100 | 55 | 78 | 100 | 100 | 47 | 70 |
That in spite of the meagre chemotherapeutic results so far obtained, the pyrvanal model possesses a greater specificity than any of the systems described by Snell,229 is shown by the following: It will only attack cysteine and cysteinyl peptides but will not degrade glutathione, urease, homocysteine, cysteamine, penicillamine or thioglycollic acid. In presence of other amino-acids, such as alanine, which could undergo transamination reactions, the hydrogen sulfide evolution from cysteine is unaffected, however, serine reduces this by about 20%. Pyridoxal is very much less effective than either pyridoxal phosphate or 5-deoxypyridoxal as a partner in the pyrvanal system. As far as it is known at present,33 the mechanism of action consists of the formation of a thiazolidine derivative (Fig. 26). This ring opens under the influence of vanadium, exposing the sulfhydryl group which is then eliminated as H2S, perhaps from a vanadium chelate similar to those proposed by Snell 229 for other metals. When versene (EDTA) was added to the mixture, both steps, ring opening and hydrogen sulfide evolution, were prevented. As pointed out above, further studies should disclose whether all this applies also to events under biological conditions.
Continue to:


