Zinc
Description
This section is from the book "The Engineer's And Mechanic's Encyclopaedia", by Luke Hebert. Also available from Amazon: Engineer's And Mechanic's Encyclopaedia.
Zinc
A metal of a bluish-white colour; of considerable hardness, and so malleable, when pure, as not to be broken with the hammer, though it cannot be thus much extended. It is, however, easily rolled into sheets, by the flatting-mill; and in this state, it has recently been brought into very extensive use, in the manufacture of pipes, gutters, and a great variety of vessels and utensils. Its specific gravity is 7.0. In a temperature of from 210° to 300°, it possesses so much ductility, that it can be drawn into wire as well as laminated, for which process, a patent was granted to Messrs. Hobson and Sylvester, of Sheffield.
Zinc melts at about 700° Fahr.; by continuing to raise the heat, it soon after becomes red hot, and burns with a dazzling white flame, and oxides with such rapidity, as to fly up in a floculent form, which is called the flowers of zinc. When these are urged by a strong fire, they are converted into a clear yellow glass.
In the ordinary method of preparing the sulphate of zinc, or spelter, the ore is exposed to the heat of a furnace, in a melting-pot, from the bottom of which, a pipe descends into a vessel of water, for the purpose of receiving the metals as they are melted, and for condensing such portion as pass off in the form or vapour. An improvement upon this mode was the subject of a patent, about fifteen years ago; in which it Was so arranged, that as the zinc volatilized, the vapour should be received and condensed in a separate vessel, leaving the melted metal, such as lead, and other impurities, in the former. An improvement upon this mode of operating, was lately patented by Messrs. Benecke and Shears; their process consists, first, in a peculiar treatment of the ore, previous to its introduction into the furnace; and secondly, in a peculiar arrangement of the retorts, and other appendages, by which a more convenient mode of charging the retorts is obtained, and a purer metal is the result.
The ores are first to be roasted in the ordinary way, by stratifying them with fuel, and setting fire to the pile. The ore is next spread out in the air, and lixiviated, to separate the sulphate of zinc; it is next to be dried, pulverized, and roasted a second time, until the sulphur is extricated, when it should be powdered again, and mixed with an equal quantity of carbonaceous matters, such as powdered coal, charcoal, cinders, etc.; in this state, it is to be saturated with an alkaline ley, or a solution of common salt; the solutions varying according to the nature of the ore. Calamine, or other oxides of zinc, will require only to be pulverized and calcined.
With the ores prepared as before mentioned, the retorts are to be charged one of those is shown in perspective at a in the annexed figure; they are made of fire-clay, or of such earth as will best stand the heat of the furnace. To the front end of these retorts are two apertures: the upper circular, for the reception of the neck of an earthen head-piece b; the lower, d, is square, for clearing out the residuum after working, which is closed during the distillation, by a stopper, and luted. The head-piece has likewise another tube fitted to it, and luted, merely for the purpose of lengthening it sufficiently to allow the vapour to cool as it descends, and to condense upon an iron plate beneath, as shown in the lowest figure in the diagram, which represents a cross or vertical section of a reverberatory furnace, in which a double row of such retorts may be supposed to be arranged, with a long aperture between them, where the fire is situated, which rests upon a grating over an arched passage that communicates with the open air outside the building; this arched passage has a door, by the opening and shutting of which the heat of the furnace is regulated as may be required; and through one of the ends of this long passage an aperture is made for supplying the furnace with fuel.
The earthen head-pieces b, it will be observed, have an aperture supplied with a stopper; through these openings the ore and carbon, prepared as before mentioned, are introduced in sufficient quantity, by means of a ladle, into the body of the retort, when the apertures are closed and luted. The operation of distillation then commences; the zinc, which rises in vapour, passes into the head-piece b, down the pipe c, and falls upon the iron plates beneath, in a condensed state.
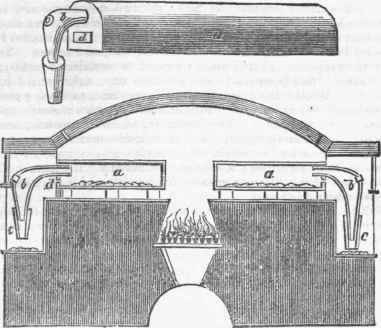
By the arrangement described, the heads and necks of each retort are placed in a square recess or neck by itself, by walls built out between them, so that each may be perfectly closed in by a door from wall to wall. The doors are made of latticed wire work, for the purpose of holding clay when plastered over them, for the purpose of effectually confining the heat within the furnace; each of these doors has a central eye-hole, provided also with a stopper, for watching the progress of the operation, and for enabling the workmen to determine the degree of heat to be applied, and other circumstances. By another arrangement the before-mentioned patentees propose to erect furnaces with several tiers of cylindrical retorts, placed one above another, with their necks or heads projecting beyond the front wall. The fire-place is covered by a low arch, to prevent the fire acting too violently upon the lowermost vessels; but through the arch apertures are made for the circulation of the heated air among the vessels above.
The carbonate of zinc, which is employed as a white pigment, is manufactured by pouring into a solution of zinc, in sulphuric acid, a solution of carbonate of ammonia, and afterwards washing and drying the precipitate. The next important use of zinc is in the fabrication of those useful and beautiful alloys with copper, called brass, prince's metal, etc. See Alloy, Copper, Brass, etc.
Blende is the native sulphuret of zinc the two substances are, however, difficult to combine artificially. The diluted sulphuric acid dissolves zinc, giving out much heat to the solvent, while hydrogen escapes. An undissolved residue is left, which Proast says, is a mixture of arsenic, lead, and copper. The white vitriol, or white copperas, as it is usually termed, is crystallized rapidly, resembling loaf sugar. Sulphurous acid also dissolves zinc, sulphuretted hydrogen being evolved. Diluted nitric acid rapidly dissolves zinc, producing much heat, with the extrication of nitrous gas. Muriatic acid operates violently upon zinc, disengaging much hydrogen. The phosphoric, fluoric, carbonic, acetic, succinic, benzoic, oxalic, tartaric, citric, and other acids, operate upon zinc, with various energy. The zinc is precipitated from its acid solutions, by means of the alkalies and soluble earths; the former re-dissolving the metal, if they be in excess. Most of the alloys or metallic combinations with zinc, have already been noticed under other heads.
Continue to:


