Renard Battery. Part 2
Description
This section is from the book "American Library Edition Of Workshop Receipts", by Ernest Spon. Also available from Amazon: American Library Edition Of Workshop Receipts.
Renard Battery. Part 2
If it is desired to exhaust the battery in a very short time, the local action, which is proportional to the time, will have a very slight influence, but the electrical rendering will be feeble, as the difference of potential at the terminals is low when the battery is giving a large discharge upon a circuit with hardly any resistance. If, on the contrary, the discharge is small, the electric rendering will be excellent, bat the local action will then become preponderant, and will diminish the total rendering. It will be understood that between these two eitremee there is a certain discharge which corresponds to the maximum rendering. Experience has demonstrated that such rendering is maximum when the difference of potential is 1.20 to 1.25 Tolt per element, whatever be [he temperature and the degree of attenuation of the liquids. This normal potential corresponds to a normal current which characterises the element. This normal current is itself a function of the temperature. Thus, for a variation of 20° C, the intensity passes from 1 lo 1.6 for the non-attenuated liquid, and from 1 to 1.4 for the liquid attenuated to 80 per cent. So it is expedient to modify the degree of attenuation of a liquid with the temperature of the season.
A liquid attenuated to 80 per cent., which is excellent daring the summer, should be replaced by a 50 per cent, liquid. for winter, when the battery is to be used for electric lighting.
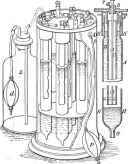
Pneumatic batter; fur electric lighting.

Light chlorochromic batter;.
A, collection of 13 elements, weighing 10 kilogrammes and having a power at no waits; B, mounting of the batter; C, platinised plate of silver rolled Into a lobe; D, zinc.
In the discharge of a battery of 24 elements in tension, containing in all 6.3 litres of liquid attenuated to 80 per cent, and discharged npon three Swan lamps of 27 volts, and of 1.25 to 1.30 ampere, mounted in derivation, the power, after a very marked syncope, at first increases regularly for an hour and a half, and afterwards slowly decreases. At the end of 2 1/3 hours the electric power disposable is insufficient to supply the three lamps. It afterwards falls very rapidly.
Taking, for a basis, the duration of 2 1/3 hours, the total electric energy produced represents 547 watts-hour - say, 35 watts-hour per litre. We can depend in practice upon 50 watts-hours per litre - say, 20 litres per kilowatt-hour. Upon constructing the different parts of the battery with care, it is possible to produce apparatus that weigh but 86 lb. per electric horse hour - say 88 lb. per kilowatt-hour, zincs included. By forcing the proportion of chromic acid, it has even been possible to obtain one horse hour for 55 lb. - say one kilowatt-hour for 70 lb. With this rich liquid, a group of 12 elements, mounted by twos in tension and by sixes in derivation, weighed 22 lb. along with the frame. Each of these groups was capable of discharging 22 kilo-grammetres (220 watts) per second at the end of 30 minutes operation - say 22 watts per kilogrammetre. Taking into account the performance of the motor, it requires four such groups, weighing 88 lb. altogether, to produce an effective power of one horse (736 watts) disposable upon the shaft.
By reducing the dimensions of the elements, Renard has succeeded in constructing a battery of 36 elements, 20 mm. diameter, weighing 11 lb., and yielding as much as 1.5 horse for 20.25 minutes, say 22 lb per electric horse, and 55-66 lb. per horse hour.
These figures establish the fact that chromochloric batteries are the highest generators of electric energy now known Despite the high price of the products used in them, they are capable, of finding an application in all cases' where lightness constitutes the main desideratum.
Selby's
In seeking for a form of cell by which the expense and internal resistance of porous pots could be avoided, I arrived at a stoneware jar containing the usual bichromate solution plus sulphuric acid, a piece of thin platinum wire clothed with pure black rubber, and a 1/8-in. tube cemented to the wire at 1 in. from the bottom. The square inch of wire was then made into a flat spiral capable of picking up a small quantity of mercury (Fig. 95), the rubber tube being simply for insulation, so that the platinum should not affect the result. On plunging this into the jar of solution, a good current was produced for a short time; when withdrawn for examination, it was found that part of the mercury still adhered to the platinum wire, although action had ceased; a fresh dip of mercury restored the effect. On investigation I found that the mercury had been used with zinc, and was in fact an amalgam. The absence of zinc meant absence of effect. On this result a simple and effective cell was formed (Fig. 96). A small jar (2 oz.
Liebig's extract of meat jar) was divided off by a glass partition cemented in with bicycle tire cement (more expensive than Chatterton's, but much stronger for grooveless partitions, jointing parchment paper cells for Leclanches, rubber, gutta-percha, shellac, and bitumen) reaching nearly to the bottom of jar; mercury is then poured in (dotted in Fig. 96) to form a "sealed joint" to separate the fluids; carbon on the one side of the partition (but suspended so as not to touch the surface of the mercury) in bichromate potash sulphuric acid solution, and on the other side zinc standing in the mercury in plain water to assist in counterbalancing the bichromate fluid on the other side, so as to require less mercury to make a safe joint. Of course the jar is represented wider than is really the case, so as to prevent confusion in the sketch. I was led to this form by many circumstances, among others by the peculiar way in which a cine in bichromate battery was "undermined" where it was outwardly protected by a cement which resisted the action of the dilute acid solution in the poroua pot.
Continue to:


