VII. - Beer. Continued
Description
This section is from the book "Alcohol, Its Production, Properties, Chemistry, And Industrial Applications", by Charles Simmonds. Also available from Amazon: Alcohol: Its Production, Properties, Chemistry, And Industrial Applications.
VII. - Beer. Continued
The first "original gravity" tables were constructed in the year 1847 by Messrs. Dobson and Phillips, of the Inland Revenue Department. They were drawn up with considerable care; but some discussion having arisen, the whole matter was referred a few years later to Profs. Graham, Hofmann, and Redwood, whose report was presented to the authorities in 1852, and published the same year in the Journal of the Chemical Society.1 A new table was drawn up, and subsequently incorporated in the Schedule to the Inland Revenue Act. 1880. An exhaustive revision of this table was carried out during the years 1909.10 by Sir Edward Thorpe and Dr. Horace T. Brown, the work including, not only laboratory experiments, but an extensive series of observations at representative breweries, in order to obtain data which should correspond fairly with the actual working conditions met with in modern brewing practice. The revised table, of which a copy is given on p. 470, was legalised by the Finance Act, 1914, for use in determining the original specific gravity of beer in the United Kingdom.2
Process, (l). Distillation method. - Most of the carbon dioxide is first expelled from the beer - which is readily done by pouring it backwards and forwards from one vessel to another in such a way as to make it froth. Where many samples are examined, a small stirrer or whisk actuated by an electric motor is convenient for the purpose.
Worts or unfinished beers containing a sediment of yeast, etc., are required to be filtered.
A convenient quantity of the beer - 75 c.c. will suffice - is measured out at 60° F. and placed in a distilling flask, the measuring flask being rinsed out with about 20 c.c. of water and the rinsings added to the main quantity, which is then distilled. The distillate is received in the original 75 c.c. flask until about four-fifths (60 c.c.) has been collected; its temperature is then adjusted to 60° F. and its volume made up to 75 c.c. The specific gravity of the distillate, referred to water as 1000, is then determined with a 50 c.c. or (preferably) a 1000.grain pyknometer, and the result subtracted from 1000. The difference is termed the "spirit indication," and the corresponding number of "degrees of gravity lost during fermentation" is found from the accompanying table (Table I, p. 470).
1 Vol. 5, 229.
2 For a fuller account of the above subject, and for a valuable study by Dr. Brown of the scientific principles involved, see the J. Inst. Brewing, 20, No. 7 (Vol. 11, new series), 1914.

Fig.'45. - still. Used for determining the original gravity of beer.
Meanwhile, the residue in the distilling flask is cooled and washed into another 75 c.c. flask, the temperature adjusted to 60° F., the volume made up to 75 c.c. with water, and the specific gravity determined. This is the "extract gravity." Adding to this the "degrees of gravity lost," as above determined, we get the original specific gravity of the beer or wort.
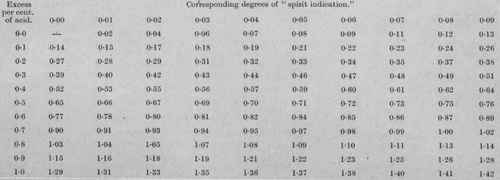
Where, however, the acidity of the beer is greater than 01 per cent., calculated as acetic acid, an additive correction is required, to compensate for alcohol which has been oxidised to acetic acid. In the official method the acidity is determined with a standard solution of ammonia, using litmus paper as indicator. From the result, expressed in terms of acetic acid, and as a percentage of the sample, 01 is deducted, and the remainder, by reference to the annexed table (Table No. III, p. 472) gives the corresponding correction of the "spirit indication."
Specific gravity of distillate................................................ | 988.45 |
,, ,, extract ............................................. | 1022. 25 |
Then "spirit indication" = 1000 . 988.45 = 11.55. |
From Table No. I, this spirit indication = 52.60 degrees of gravity lost. Hence, if the beer contains no excess of acid,
Original gravity == 5260 + 102225 = 107485.
If, however, the beer contains, let us say, 035 per cent. of acid, calculated as acetic acid, then the excess, after deducting 01, = 025, which from Table No. III - 033 degree of spirit indication. Adding this to the actual indication, we get 11.55+0.33 = 11.88, the corrected spirit indication, which = 5425 degrees of gravity lost. Whence the original gravity = 54.25 + 1022.25 = 1076.50.
The 01 per cent. acidity was taken as the average acidity of unfermented wort in Profs. Graham, Hofmann, and Redwood's experiments. It may be noted that fresh wort contains practically no acetic or other volatile acid: the acidity is chiefly due to lactic acid.
When the proportion of volatile acid is very high, it affects the specific gravity of the distillate and so prevents the accurate determination of the spirit indication. In this case, the distillate should be neutralised with solution of sodium hydroxide and re. distilled. The determination of the acetic acid when the proportion is high is best done by distillation in a current of steam as described for wine. Alternatively, the total acidity of 50 c.c. may be determined with standard ammonia and litmus paper as usual, and the fixed acidity on a similar quantity after evaporation to dryness on a steam-bath, and redissolving in water: the volatile acidity is then given by the difference.
The "standard " ammonia may be either of decinormal strength, or of sp. gr. 9986. In the latter case, each c.c. used represents 01 per cent. of acetic acid when 100 c.c. of beer are taken for titration.
(2). "Evaporation" method. - In this process, it is not necessary actually to distil the beer. The specific gravity of the beer, freed from carbon dioxide as already explained, is determined, and then 75 c.c. or other convenient quantity, measured at 60° F., is evaporated by gentle boiling in a beaker or basin until the volume is reduced to about one-third. During the operation care must be taken not to char any of the solid matter. The alcohol being thus expelled, the residue is cooled, made up at 60° F. with distilled water to the original volume, and the specific gravity taken: this is the "extract gravity."
From this "extract gravity" deduct the specific gravity of the beer; the difference is the "spirit indication " for the evaporation method. Referring this to Table No. II we get the corresponding " degrees of gravity lost," which, added to the "extract gravity," furnishes the original gravity required. The correction for excess acidity is made in the same way as in the distillation method.
Example: Sp. gr. of the beer, 100925. Extract gravity, 1021.48. Total acidity, 0 26 per cent.
Then spirit indication = 1021.48 - 100925 == 1223. Excess acidity = 0.26 - 0.1 = 016, which from the table (No. III) gives 0.22 correction of spirit indication;
.. corrected spt. indn.= 12.45 = 5805 degrees of gravity lost, from Table No. II.
Whence the original gravity = 1021.48+58.05 = 1079.53.
As compared with the distillation method, the evaporation process is somewhat the quicker, and does not require the use of a still. But the Table No. II has not received the thorough revision which Table No. I has undergone; moreover, the distillation process is the one legalised for use in this country.
It may be noted that if a sufficiently sensitive hydrometer be used for taking the specific gravities, instead of a bottle, the original gravity of beer can be determined by the evaporation process without the use of either a balance or a still.
For rough purposes, it is convenient to remember that 1 per cent. of proof spirit produced in fermentation corresponds with about 3.5 degrees of gravity lost.
Table No. I. - Distillation process.
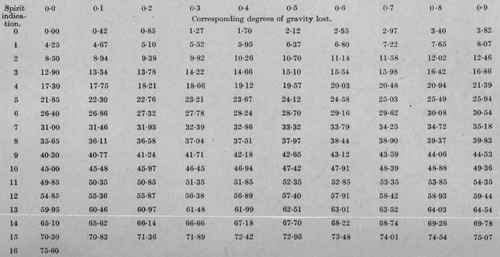
Table No. II. - Evaporation process.

Table No. III.
For ascertaining the indication value of the acetic acid.
Continue to:


