Chapter III. The General Chemistry Of The Alcohols
Description
This section is from the book "Alcohol, Its Production, Properties, Chemistry, And Industrial Applications", by Charles Simmonds. Also available from Amazon: Alcohol: Its Production, Properties, Chemistry, And Industrial Applications.
Chapter III. The General Chemistry Of The Alcohols
For a complete account of the important class of bodies termed "alcohols," including the proofs of their chemical constitution, reference must be made to text-books of organic chemistry. It would be beyond the scope of the present work to set out fully the reasons which have led chemists to group together as "alcohols" substances so dissimilar in appearance as light, mobile wood spirit; viscid, heavy glycerol; and solid, wax-like cerotin. None the less, it will be useful and convenient to have here a summary of the chief chemical facts which are common to the alcohols as a whole.
In this chapter, therefore, it is proposed briefly to describe the chemical characters which distinguish the alcohols as a class, and more particularly the aliphatic, monohydric alcohols. The special properties of ethyl alcohol, as also of its congeners methyl, propyl, butyl, and amyl alcohols, will be dealt with in subsequent chapters.
Ordinary alcohol (ethyl alcohol, C2H5OH) is the best-known member of a series of neutral substances, some liquid, others solid, which can be looked upon chemically as derived from hydrocarbons by the replacement of one hydrogen atom by a hydroxyl group.
Thus corresponding with: -
Methane, | CH4, | we have | CH3.OH, | methyl alcohol, | ||
Ethane, | C2H6, | " | C2H5.OH, | ethyl | " | |
Propane, | C3H8, | " | C3H7.OH, | propyl | " | |
Butane, | C4H10, | " | C4H9.OH, | butyl | " | |
Pentane, | C5H12, | " | C5H11.OH, | amyl | " | |
And so on; the further members of this series of alcohols being designated hexyl, heptyl, octyl, etc., alcohols, according to the number of carbon atoms which they contain.
Classification
Alcohols are classified as monohydric, dihydric, trihydric, and polyhydric, according to whether one, two, three, or more hydroxyl groups are present in them. Thus those of the series represented above are all monohydric alcohols. Glycol, the formula of which is C2H4(OH)2, is an example of a diydric alcohol; and glycerol, C3H5OH)3, is a trihydric alcohol. In this work, we are concerned almost exclusively with the monohydric series.
A further distinction into primary, secondary, and tertiary alcohols is based upon differences of structure. When a carbon atom is linked to only one other carbon atom, it is termed "primary. If linked to two others, it is a secondary, and if to three, a tertiary, carbon atom. Now a primary alcohol is one in which the hydroxyl group is attached to a primary carbon atom; whilst in a secondary or a tertiary alcohol the hydroxyl group is attached to a secondary or a tertiary carbon atom respectively. Whence it follows that: -
1. Primary alcohols contain the group -CH1OH. Example: CH3CH1CH1OH, propyl alcohol.
2. Secondary alcohols contain the group
Example:
C3CH(O).CH3, isopropyl alcohol.
3. Tertiary alcohols contain the group tertiary isobutyl alcohol.
Example:
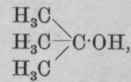
Methyl alcohol, CH3-OH, is a primary alcohol containing only one carbon atom. The group -CH1.OH is in this case attached to a hydrogen atom.
Continue to:


