Distillation
Description
This section is from "The American Cyclopaedia", by George Ripley And Charles A. Dana. Also available from Amazon: The New American Cyclopædia. 16 volumes complete..
Distillation
Distillation (Lat. destillare, to drop), the conversion of a liquid or a solid into a vapor and condensing it; usually applied to liquids. If sea water is boiled, the vapor which passes off leaves behind the salts and other substances held in solution, and by condensation in the atmosphere, or against cool surfaces, is converted into drops of pure distilled water. By artificial processes of a similar nature a volatile liquid may be separated from one less so, as alcohol, acetic acid, or ether from water with which they may be mingled. The volatile principles of plants may be extracted by water or other liquids, and by distillation separated in a pure state, or dissolved in the liquid used for extraction. Solid vegetable and animal substances, by exposure to heat in vessels more or less closed, undergo a process in which the organic compounds are destroyed, and their constituents recombined, partly in the form of volatile products, which may be collected as distillates, and partly as non-volatile residuum. By subjecting wood to such a process, charcoal, tar, pyroligneous acid, and naphtha are produced; and by heating bituminous coal in close crucibles, illuminating gas, coke, coal, naphtha, and other products are separated.
When solid substances, such as sulphur or camphor, are volatilized and condensed, the process, strictly speaking, is not distillation, because there is no collecting in drops; but it is called sublimation. In the chemical laboratory distillation is commonly conducted in glass retorts and receivers, the boiling taking place in the retort and the condensation in the receiver, which is usually cooled by the application of water or ice to the exterior. The simplest apparatus of this kind is shown in fig. 1.

Fig. 1. - Simple Distilling Apparatus.
When large products are required, more efficient forms of apparatus must be employed. Such are stills, in which the retort as well as the condensing portion is made of metal, generally copper or iron. Usually, instead of a receiver of the ordinary form, the neck of the retort, or a tube from the boiler, is converted into a long coil, called a worm, which, being immersed in a tub of water, causes condensation of the vapors within. A simple form of still, often used when considerable quantities of distilled water are required, and also for other pharmaceutical purposes, is represented in fig. 2. The retort is all that portion of the apparatus which sets in and upon the furnace, including the neck c, terminating at d, in the worm d, d, which passes through a cooler e, which is supplied with cold water by the funnel h, the water entering at the bottom and flowing out at the top. It will be seen that the form of this apparatus will prevent some of the less volatile portions of the liquid subjected to distillation from passing over, as they may with the application of much heat in the common retort in fig. 1, a considerable condensation taking place in the dome b.
Liebig's laboratory distilling apparatus is constructed by passing the neck of a retort into a Liebig's condenser, which consists of a glass tube surrounded by a metallic one, of tin or copper, in the lower part of which water enters, flowing out at the top. The arrangement is on the same plan as the still in fig. 2, only instead of a worm there is a straight tube. - The preparation of an alcoholic liquor by separating the more volatile portions of the fermented juices of fruits and infusions of grains does not appear to have been understood by the ancients. Ure says: "It seems to have been invented by the barbarians of the north of Europe as a solace to their cold and humid clime, and was first made known to the southern nations in the writings of Arnoldus do Villa Nova, and his pupil, Raymond Lully of Majorca." But there are now few nations above the condition of savages who do not prepare some kind of alcoholic liquors by distillation. The fermented juices of the grape and other fruits, and the fermented infusion of grape sugar derived from malted liquors, contain the same intoxicating principle, alcohol, which it is the object of distillation to obtain in a more condensed form, and which when so obtained from liquids of different qualities retains the peculiar aroma and flavor of the plant, until by repeated distillations and rectifications the pure alcohol is at last obtained from peculiar volatile oils or flavors.
All the juices of plants which can undergo vinous fermentation, and all vegetable matters which contain starch, may thus be made to produce alcoholic liquors. Some animal fluids also, which contain saccharine matters, as milk, may be made to furnish alcohol by fermentation and distillation. An intoxicating liquor from this source, called kumiss, is made in Tartary, both simply fermented and distilled. The fruits of each country furnish spirits of their peculiar flavors when these are obtained directly from the fermented juice; -but if this is first allowed to crystallize, the sugar so obtained, on being redissolved and fermented, is found to have lost the aroma of the plant. Thus, the high flavor of the rum which is distilled from fermented fresh cane juice is not found in the distillate from fermented sugar and molasses. Sugar-growing countries produce rum, vine-growing countries brandy, and grain-growing countries whiskey and gin. The Chinese manufacture a distilled liquor from rice, and the inhabitants of Kamtchatka another from mushrooms.
The processes of obtaining these liquors are essentially the same, except that the cereals require some preparatory operations before they are ready, like the saccharine juices, for fermentation, and these operations are almost identical with those employed in brewing ale; the only difference being that in preparing the wort which after fermentation is to be distilled, the action of the diastase in the malt is continued until the dextrine is transformed into grape sugar. The grain is subjected to the process of mashing, and the resulting wort to that of cooling and fermentation, after which follows the distillation. - A brief account of the manufacture of whiskey, as performed in the great distilleries of Scotland, will sufficiently explain the various operations. Barley is commonly used as the starchy material, and is more or less mixed with oats, rye, or other grains. It may be malted wholly or in part, or may be used with sugar. Barley malt is the best material, but the heavy duty imposed upon it restricts its use.
According to Dr. Thomson, 40 bushels of ground barley are mixed with 20 bushels of bruised malt, in a mash tun of cast iron, together with about 750 gallons of water, at a temperature of about 150° F. The mashing is continued one hour and a half, during which time 500 gallons more of water at 190° to 205° are introduced at intervals, to keep up the heat. The whole is then allowed to infuse two hours, during which time the grain subsides, and the liquid above it is a saccharine turbid fluid, called wort, which also still contains some starch and dextrine; but these by the action of the diastase are gradually converted into grape sugar. At the end of the two hours' infusion the greater part (usually about two thirds) of the wort is drawn off, and 500 gallons of water at 190° is added, and the infusion is renewed and continued another hour and a half. After the wort is again drawn off, a third infusion succeeds, with 800 gallons of boiling water. This being well stirred for 20 minutes, and then left about half an hour, the saccharine matters are found to be extracted. The weak wort is then drawn off and boiled down to the required strength, or it is added to the first and second worts, or is kept to be used instead of pure water for the first infusion of the next mashing.
Strong worts are not desirable, the fermentation being more complete and the yield of spirits greater . when they are of moderate specific gravity. By the old excise laws of Great Britain they were required to be of a certain high degree of strength, but in Scotland and Ireland they are now allowed to range from 1030 to 1080 sp. gr., water being 1000. The next process is that of cooling the worts, and in consequence of the tendency of those produced from raw grain to become acid, this must be rapidly accomplished. In some distilleries the wort is run into large shallow coolers in airy situations in the upper part of the building. In others the cooling is done by a more compact form of refrigerator, a description of which is given in the article Brewing. The temperature is usually reduced to between 70° and 75°, when the worts are transferred to the fermenting tuns, and yeast is added in the proportion of about one gallon to 100 gallons of wort. The object of fermentation is to convert all the saccharine matter, if possible, into alcohol and carbonic acid; but the presence of the alcohol as it is formed impedes the progress of this change, and a quantity often amounting to one fifth of the whole saccharine matter escapes decomposition.
By the invention of Mr. Sheridan in fermenting the wash in close tuns, and causing the alcohol to evaporate by using a powerful air pump, the whole saccharine matter was converted into alcohol; but the excise restrictions prevented the adoption of the improvement. As the fermentation proceeds the liquor attains a less specific gravity, and when successfully conducted its density gets to be the same as that of water. If it is pushed too far, or goes on sluggishly or at too high a temperature, loss will result by a portion of the alcohol passing to acetic acid, the presence of which is indicated by increased specific gravity, as well as by its peculiar odor and taste. The process of distillation, which, by distinguishing the preparation of ardent spirits from that of fermented liquors, gives its name to the whole operation, now succeeds the fermentation. It is conducted in stills of various sizes and forms, some of which have a capacity equal to distilling from 2,000 to 3,000 gallons of wash per hour.
The origin of the first still which abolished to a great extent the use of the worm and substituted condensing vessels, which principle has been retained with modifications in nearly all subsequent inventions of the kind, is due to a Frenchman named Edou-ard Adam, who is said to have been a distiller, unacquainted with anything more than the routine of his trade.. In 1801 he witnessed some experiments with a "Woulfe apparatus at a chemical lecture in Montpellier, and was so impressed with its advantages that he soon after constructed a still upon the same principle. This succeeded so well that the whole-process of distillation was soon completely changed. The uses of Woulfe's apparatus are described in the article on Gas, and by referring to that it will be seen that Adam's still was one of the happiest adaptations of a laboratory appliance to a manufacturing purpose. The modification of it as made by M. Adam is represented in fig. 3. A number of egg-shaped copper vessels, corresponding to the Woulfe bottles, are placed in convenient situations near each other, the terminal ones being connected on the one hand with the retort or boiler containing the fermented liquor or wine, and on the other with a worm which is immersed in a cooler.
The neck of the retort passes into the first egg-shaped vessel, dipping below the surface of the liquor. It is perforated at its termination with minute holes through which vapor passes. A pipe from the first egg leads to the second, also dipping beneath the surface of the liquor, and so on, from one to the next, whatever the number may be. From the last egg a tube enters the globe B before passing into the worm, whose use will presently be explained. From next to the last egg, or from any one of the series, an extra tube,' 0, also passes into the globe B, by which arrangement one or more of the eggs may be dispensed with when the distillation does not need to be carried very high. Another pipe, D, connects each egg and also the boiler with a small worm, V, which is used for testing the strength of the distillate in any one of the eggs, or from the boiler. Another pipe, E, leads from the cooler F into the boiler, and another, H, into the cooler from the storehouse where the wines are kept. The worm in the cooler F, moreover, leads into another worm in the cooler G. This still is worked in the following manner: The cocks connecting the upper tubes are closed, and those in the lower pipe, E, are opened.
The wine is pumped from the storehouse through the tube H into the cooler F, whence it flows into the boiler. When this is about two thirds full the cock next it is closed, and the wine is forced up into the first egg; when this is about half tilled the cock next it is closed, when the second egg is treated in the same manner; and so on through the series, except the last one, which serves as a condenser and is surrounded with cold water. The lower cocks are now closed, and the upper ones communicating between the eggs, and with the worm, are opened. Heat is applied to the boiler, and the mixture of alcoholic and watery vapor is carried into the first egg, and there condensed by the wine, quite rapidly in the beginning of the process, so that for a time no vapor passes over into the second egg. The wine in the first egg, however, gradually comes to its boiling point, which, by reason of its containing more alcohol than that in the boiler, is at a lower temperature. In consequence, the vapor which passes into the second egg has a greater percentage of alcohol than that which it received. This vapor, being condensed, will cause the liquor in the second egg to be stronger than in the first, and therefore to boil at a still lower temperature.
The successive eggs, as they recede from the boiler, will thus contain stronger and stronger spirits, so that the last one may be made to receive alcoholic vapor of any desired strength. This is passed into the worm in F and condensed either in that or in the succeeding worm in the tub below, which is filled with water kept cool by a constant flow. The upper cooler, containing the wine, is kept I closed, except that a pipe leads into the globe B. This arrangement is for the purpose of preventing loss of spirit by evaporation, which would be considerable at the temperature it attains by contact with the worm. The excise laws of Great Britain prevented the introduction of this still into that country until after their modification in 1815. Adam's apparatus was in the mean time much improved in France by Isaac Berard, Cellier-Blumenthal, and Derosne. The modification of Cellier-Blumenthal, im-proved by Derosne, and now called Derosne's still, is represented in fig. 4. It consists of two boilers, A, A'; a first rectificator, B; a second rectificator, C; a wine heater, D, containing a dephlegmator; a condenser, F; a supply regulator, E, for controlling the flow of wine from the reservoir G, which is accomplished by means of a floating ball.
The still is worked in the following manner: The boilers are about two thirds filled with wine or the liquor to be subjected to distillation through the cocks c, c'. The proper quantity is indicated by the glass gauges d, d'. Wine from the reservoir G is then let into the funnel J, by which the condenser F and the wine heater D are filled. On the application of heat the low-wine vapors pass from the lower into the upper boiler through the pipe Z, the extremity of which is enlarged and perforated with small holes. Here the vapors are condensed, increasing the strength of the wine in the upper boiler, and consequently lowering its boiling point. The vapors ascend into the rectificators B and C. The lower rectificator, B, contains a number of shallow pans perforated with holes, and a number of spherical disks, also perforated with holes, placed above them, in pairs, the convexity of each disk being upward, and receiving the drip of the shallow pan next above it. This drip is produced by warmed wine which flows from the wine heater through the pipe L. By. these means the vapors ascending from the upper boiler have their more watery portions condensed, while the alcoholic vapor continues to ascend.
The dripping wine also has a portion of its alcohol expelled in the form of vapor, which ascends with the vapor coming from below into the upper rectificator through the orifice O in its bottom. This upper rectificator communicates through the tube M with a worm (which is the dephlegmator) in the wine warmer D, the worm ending in the tube m, which again terminates in the worm contained in the condenser F through a cylindrical connection in its upper part. The worm in F terminates in a small vessel, N, which is furnished with an alcoholometer. The alcohol in N flows from its upper part into the cistern II. The upper rectificator 0 is divided into a number of compartments by as many horizontal partitions, each disk having an orifice in its centre, like the orifice at O. To each of these orifices on the upper side of the partition is adjusted a short open vertical tube. A short distance above each tube is placed an inverted pan, having its edges descending about three fourths of an inch below the level of the upper orifice of the tube. As the vapors ascend from the lower rectificator into the upper one, a portion of them condense and collect upon the bottom of the compartments until they rise slightly above the edges of the inverted pans and nearly to the upper orifices of the tubes.
When this takes place the vapor can only pass upward by forcing its way under the edges of the pans, by which means the more watery portion is still further condensed, the more alcoholic vapor, having a higher tension, retaining its gaseous form, and passing on through the tube M into the dephlegmatory worm in the wine heater, there to be partially condensed; which process heats the wine surrounding the worm. A phlegma collects in the lower convolutions, which may be drawn off by means of the pipes p, p, p, and transferred at pleasure either into the tube m or into the upper rectificator. The purer alcoholic vapors which arise pass through the dephlegmator into the condensing worm in the condenser F, whence they flow in liquid form into the vessel N, and thence into the cistern H. The strength of the alcohol produced by this still depends upon the number of windings of the dephlegmator, and the number of partitions in the upper rectificator. Derosne's still requires but little fuel, distils rapidly, and yields a good spirit, which may be varied in strength at pleasure; but it is rather complicated, and may with advantage, especially when spirits of only one strength are required, be replaced by a simplification of it, devised by Laugier. - The products of the distilleries of different localities are distinguished by peculiar flavors which give them a reputation; but they are not always, especially when the liquor is made from cereals, derived from the original qualities of the materials, but often from the fuel used in drying the malt.
Much of the Scotch and Irish whiskey possesses the peculiar flavor of peat smoke. Brandies manufactured from wines are made to retain their peculiar flavors by conducting the distillation at a temperature sufficient to carry over the volatile oils and ethers, and also enough water to reduce the strength considerably below that of ardent spirits produced in Great Britain; but they may be given strength by the addition of pure spirit. - Common alcohol is the hydrated oxide of ethyle, or ethylic alcohol, C4H10O+H2O = C2H6O. In the process of vinous fermentation there are formed, under conditions which are not yet clearly understood, other homologous alcohols, such as propylic, butylic, and amylic alcohols, in greater or lesser quantities. They have a higher boiling point than ethylic alcohol, and therefore do not usually appear in the first products of distillation; but in the latter stages of the operation, when the heat is raised, they pass over into the worms or condensers. These alcohols, mixed with some ethylic and small quantities of various ethers and volatile fatty acids, such as capric, caprylic, and formic, constitute an oily substance which passes under the general name of fusel oil.
That of potato spirit consists almost wholly of amylic, combined with a small quantity of ethylic alcohol. This fusel oil imparts a very disagreeable taste to spirits, and to get rid of it is often a matter of importance. The process is called rectification, and tnay be conducted with or without redistillation. Its complete removal by distillation, even with the use of alkalies and other substances, is somewhat difficult and expensive, notwithstanding that the boiling point of absolute ethylic alcohol, 173°, is much lower than that of the others, amylic alcohol boiling at 270°. It is found, however, that an elimination can be satisfactorily effected by filtration through granulated charcoal which has been recently heated. - About three fifths of the products of distillation in the United States are what are termed high-wines or whiskey, containing about 75 per cent. of alcohol. This, as it comes from the still, contains a good deal of fusel oil. Some of it is made into cheap whiskey, and the remainder is rectified and redistilled into French spirit. When the percentage of alcohol is high it forms Cologne spirit.
About one fifth of the products of distillation is alcohol, which is all used for manufacturing and mechanical purposes, about 6,000 barrels being exported from the United States monthly. The remaining one fifth of the distilled liquors consists of whiskey and rum, distilled to about proof, and so left to ripen with age. Considerable whiskey is made from rectified spirits of about 75 per cent. alcohol, which is reduced and flavored by the addition of raw whiskey not rectified. All distilled liquors are sold by the gallon, according to the proof. Proof spirit is reckoned by the government as 100, which is 50 per cent. absolute alcohol. - Destructive Distillation. When organic bodies are excluded from the air and subjected to heat, they undergo decomposition, and the constituent atoms or molecules rearrange themselves into new compounds. The causes of this are various, and depend much upon the conditions which are present. The elective affinities of atoms and molecules, when a variety of substances are mingled together, vary with the heat and with the nature of the substances with which they are surrounded.
Two or more elements united in a compound, requiring a certain degree of heat to separate them, will require less when another compound is present, the amount depending upon the affinity which a constituent of one compound may have for a constituent in the other. Organic bodies, which are usually composed of several compound constituents (as woody fibre, starch, resin, oils, and water), very readily suffer a complete change in their chemical as well as their organic structure by the simple application of heat, and without the presence of a supporter of combustion. When wood is enclosed in an iron tube and subjected to heat, various new bodies are formed of an inorganic character, and of a composition and number depending upon the duration and degree of heat. Decomposition commences at about 284° F.; and between this and red heat various gases, vapors, liquids, and solids are produced. The products of the lowest temperature contain the most oxygen, as water and carbonic and acetic acids. As the temperature rises bodies containing less hydrogen are formed, such as wood spirit, acetone, and creosote. At a still higher temperature, hydrocarbons, such as toluene, xylene, eupione, and paraffine, appear; and as the temperature approaches redness, hydrogen is abundantly formed.
By connecting the retort with condensing vessels, by means of good-sized exit tubes, the various volatile products may be collected, and subsequently separated by fractional distillation and otherwise. The manufacture of illuminating gas illustrates the destructive distillation of bituminous coal on a large scale. The products may be divided into three classes, viz.: 1, coke, consisting of carbon, sulphuret of iron, and ash; 2, ammoniacal liquor, containing carbonate, sulphide, chloride, cyanide, and sulpho-cyanide of ammonium; 3, tar, embracing a great variety of solid and fluid hydrocarbons and acids, among which are benzole, toluole, xylole, naphthaline, anthracene, and carbolic or phenic, oxyphenic, and cresylic acids, together with creosote, a compound of homologous oxyphenic and methylic acids; also several bases, as aniline, iridoline, and rubidine; 4, illuminating gas, containing light-yielding compounds, such as acetylene, C2H2; ethylene, or olefiant gas, C2H4; benzole, C6H6; naphthaline, C10H8; pro-pyle, C3H7; butyle, C4H7; mingled with hydrogen, carbonic oxide, and impurities, such as carbonic acid, ammonia, cyanogen, sulpho-cyanogen, and sulphuretted hydrogen. These processes involve highly complex reactions, the many stages of which are yet imperfectly understood.
The destructive distillation of acids and the simpler bases are more easily understood, the products being often readily traced to their origin. The decomposition of the acids takes place differently according as they are separate or in the presence of bases. When distilled alone many undergo a simple elimination of carbonic acid, with the formation of a pyro-acid. Thus, gallic acid, when heated in a close vessel to 419° F., is decomposed into pyrogallic and carbonic acids, as follows: c7H6O5 = c6h6o3 + co2

Fig. 2. - Still.
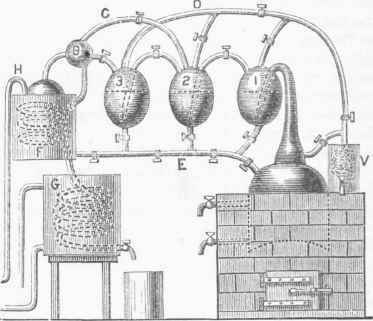
Fig. 3. - Edouard Adam's Still.
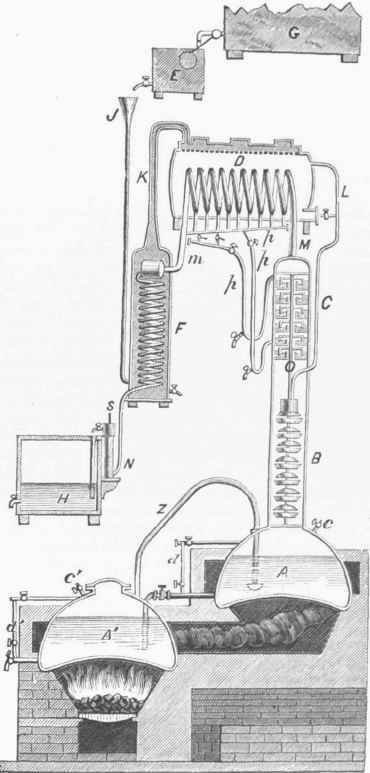
Fig. 4. - Derosne's Still.
Gallic acid. Pyrogallic acid. Carbonic acid.
Other reactions are not quite as simple. Thus, oxalic acid yields water, carbonic acid, carbonic oxide, and formic acid, as follows:
2C2H204 = H20 + CO + 2CO2 + CH2O2 Oxalic acid. Water. Carb. ox. Carb. acid. Formic acid.
As an example of the destructive distillation of a salt, may be taken that of acetate of lime, which is converted into acetone and carbonate of lime, thus: -
2C2H3CaO2 = Ca2CO3 + C3H6O Acetate of lime. Carb. lime. Acetone.
- Fractional Distillation is the separating of different constituents which naturally exist or have been artificially produced in a mass. This is accomplished quite readily on account of the different temperatures at which the various constituents pass into vapors. The refining of petroleum is an example of fractional distillation. (See Petroleum.)
Continue to:


